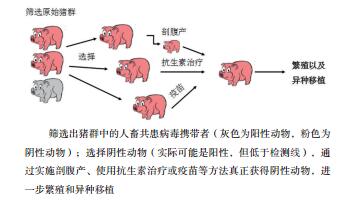
-
摘要: 异种移植中采用猪的细胞、组织和器官可能导致猪传染性病原体的转移,进而感染人受体,甚至引发人畜共患的疾病。为防止此情况发生,应对供体动物进行广泛的微生物筛选,包括细菌、病毒、真菌和其他微生物。在进行移植之前,供体猪可进行长时间的筛选,故异种移植将比同种异体移植变得更为安全。通过实施剖腹产、接种疫苗等策略可以筛选出不转移不传播微生物的供体动物。猪内源性逆转录病毒(PERV),整合于所有猪的基因组中,上述方法不能清除PERV,因此需选择病毒低表达的猪或构建PERV表达抑制的基因修饰猪。
-
表 1 已开发疫苗治疗病毒和细菌感染的疾病
Table 1. Diseases with available vaccines against viral and bacterial infection
疾病 病原体 假狂犬病 猪疱疹病毒1 手足口病 手足口病病毒(口蹄疫病毒) SMEDI综合征 猪细小病毒 猪繁殖和呼吸综合征 猪繁殖与呼吸综合征病毒 猪瘟 猪瘟病毒 猪流感 猪流感病毒 传染性胃肠炎 传染性胃肠炎病毒(冠状病毒) 坏死性胸膜肺炎 胸膜肺炎放线杆菌 萎缩性鼻炎 多杀性巴氏杆菌 梭菌病 梭菌 大肠杆菌性腹泻 大肠杆菌 地方性肺炎 猪肺炎支原体 丹毒 丹毒丝菌 格拉泽氏病 猪副嗜血杆菌 出血性败血症 多杀性巴氏杆菌 链球菌脑膜炎 猪链球菌、副猪嗜血杆菌 SMEDI综合征为死产、木乃伊产、胚胎死亡、不育综合征 -
[1] Ljungman P, Boeckh M, Hirsch HH, et al. Definitions of cytomegalovirus infection and disease in transplant patients for use in clinical trials[J]. Clin Infect Dis, 2017, 64(1):87-91. DOI: 10.1093/cid/ciw668. [2] Warren-Gash C, Childs K, Thornton A, et al. Cirrhosis and liver transplantation in patients co-infected with HIV and hepatitis B or C:an observational cohort study[J]. Infection, 2017, 45(2):215-220. DOI: 10.1007/s15010-016-0976-x. [3] Zhou H, Zhu W, Zeng J, et al. Probable rabies virus transmission through organ transplantation, China, 2015[J]. Emerg Infect Dis, 2016, 22(8):1348-1352. DOI: 10.3201/eid2208.151993. [4] Spizzo T, Denner J, Gazda L, et al. First update of the International Xenotransplantation Association consensus statement on conditions for undertaking clinical trials of porcine islet products in type 1 diabetes——chapter 2a:source pigs——preventing xenozoonoses[J]. Xenotransplantation, 2016, 23(1):25-31. DOI: 10.1111/xen.12223. [5] Wynyard S, Nathu D, Garkavenko O, et al. Microbiological safety of the first clinical pig islet xenotransplantation trial in New Zealand[J]. Xenotransplantation, 2014, 21(4):309-323. DOI: 10.1111/xen.12102. [6] Semaan M, Rotem A, Barkai U, et al. Screening pigs for xenotransplantation:prevalence and expression of porcine endogenous retroviruses in Göttingen minipigs[J]. Xenotransplantation, 2013, 20(3):148-156. DOI: 10.1111/xen.12032. [7] Morozov VA, Morozov AV, Rotem A, et al. Extended microbiological characterization of Göttingen minipigs in the contex of xenotransplantation:detection and vertical transmission of hepatitis E virus[J]. PLoS One, 2015, 10(10):e0139893. DOI:10.1371/journal. pone.0139893. [8] Denner J. Xenotransplantation and hepatitis E virus[J]. Xenotransplantation, 2015, 22(3):167-173. DOI: 10.1111/xen.12156. [9] Meng XJ. From barnyard t of ood table:the omnipresence of hepatitis E virus and risk for zoonotic infection and food safety[J]. Virus Res, 2011, 161(1):23-30. DOI: 10.1016/j.virusres.2011.01.016. [10] Pitkin Z. New phase of growth for xenogeneic-based bioartificial organs[J]. Int J Mol Sci, 2016, 17(9):E1593. DOI: 10.3390/ijms17091593. [11] Kim Y, Lee C. Ribavirin efficiently suppresses porcine nidovirus replication[J]. Virus Res, 2013, 171(1):44-53. DOI: 10.1016/j.virusres.2012.10.018. [12] Wu T, Li SW, Zhang J, et al. Hepatitis E vaccine development:a 14 year odyssey[J]. Hum Vaccin Immunother, 2012, 8(6):823-827. DOI: 10.4161/hv.20042. [13] Sanford BJ, Opriessnig T, Kenney SP, et al. Assessment of the crossprotective capability of recombinant capsid proteins derived from pig, rat, and avian hepatitis E viruses (HEV) against challenge with a genotype 3 HEV in pigs[J]. Vaccine, 2012, 30(44):6249-6255. DOI: 10.1016/j.vaccine.2012.08.013. [14] Su CA, Iida S, Abe T, et al. Endogenous memory CD8 T cells directly mediate cardiac allograft rejection[J]. Am J Transplant, 2014, 14(3):568-579. DOI: 10.1111/ajt.12605. [15] Taveira A, Ponroy N, Mueller NJ, et al. Entry of human cytomegalovirus into porcine endothelial cells depends on both the cellular vascular origin and the viral strain[J]. Xenotransplantation, 2014, 21(4):324-340. DOI:0.1111/xen.12097. [16] Whitteker JL, Dudani AK, Tackaberry ES. Human fibroblasts are permissive for porcine cytomegalovirus in vitro[J]. Transplantation, 2008, 86(1):155-162. DOI: 10.1097/TP.0b013e31817d4823. [17] Mueller NJ, Fishman JA. Herpesvirus infections in xenotransplantation:pathogenesis and approaches[J]. Xenotransplantation, 2004, 11(6):486-490. DOI: 10.1111/j.1399-3089.2004.00180.x. [18] Mueller NJ, Kuwaki K, Dor FJ, et al. Reduction of consumptive coagulopathy using porcine cytomegalovirus-free cardiac porcine grafts in pig-to-primate xenotransplantation[J]. Transplantation, 2004, 78(10):1449-53. doi: 10.1097/01.TP.0000141361.68446.1F [19] Denner J. Xenotransplantation and porcine cytomegalovirus[J]. Xenotransplantation, 2015, 22(5):329-335. DOI: 10.1111/xen.12180. [20] Kharfan-Dabaja MA, Boeckh M, Wilck MB, et al. Reanalysis of transvax immunogenicity[J]. Lancet Infect Dis, 2013, 13(1):18. DOI:1. 10.1016/S1473-3099(12)70296-7. [21] Schleiss MR. Developing a vaccine against congenital cytomegalovirus (CMV) infection:what have we learned from animal models? where should we go next?[J]. Future Virol, 2013, 8(12):1161-1182. DOI: 10.2217/fvl.13.106. [22] Pass RF, Zhang C, Evans A, et al. Vaccine prevention of maternal cytomegalovirus infection[J]. N Engl J Med, 2009, 360(12):1191-1199. DOI: 10.1056/NEJMoa0804749. [23] Burbelo PD, Ragheb JA, Kapoor A, et al. The serological evidence in humans supports a negligible risk of zoonotic infection from porcine circovirus type 2[J]. Biologicals, 2013, 41(6):430-434. DOI: 10.1016/j.biologicals.2013.09.005. [24] Baró J, Segalés J, Martínez J. Porcine circovirus type 2(PCV2) enteric disease:an independent condition or part of the systemic disease?[J]. Vet Microbiol, 2015, 176(1/2):83-87. DOI: 10.1016/j.vetmic.2015.01.006. [25] Quintana J, Segalés J, Calsamiglia M, et al. Detection of porcine circovirus type 1 in commercial pig vaccines using polymerase chain reaction[J]. Vet J, 2006, 171(3):570-573. DOI: 10.1016/j.tvjl.2004.12.008. [26] Dubin G, Toussaint JF, Cassart JP, et al. Investigation of a regulatory agency enquiry into potential porcine circovirus type 1 contamination of the human rotavirus vaccine, Rotarix:approach and outcome[J]. Hum Vaccin Immunother, 2013, 9(11):2398-2408. doi: 10.4161/hv.25973 [27] Heinze J, Plotzki E, Denner J. Virus safety of xenotransplantation:prevalence of porcine circovirus 2(PCV2) in pigs[J]. Ann Virol Res, 2016, 2(3):1023. https://www.jscimedcentral.com/Virology/virology-2-1023.pdf [28] Irgang M, Laue C, Velten F, et al. No evidence for PERV release by islet cells from German landrace pigs[J]. Ann Transplant, 2008, 13(4):59-66. http://www.annalsoftransplantation.com/reprintOrder/index/idArt/880220 [29] Yang L, Güell M, Niu D, et al. Genome-wide inactivation of porcine endogenous retroviruses (PERVs)[J]. Science, 2015, 350(6264):1101-1104. DOI: 10.1126/science.aad1191. [30] Kaulitz D, Mihica D, Dorna J, et al. Development of sensitive methods for detection of porcine endogenous retrovirus-C (PERV-C) in the genome of pigs[J]. J Virol Methods, 2011, 175(1):60-65. DOI: 10.1016/j.jviromet.2011.04.017. [31] Denner J, Mihica D, Kaulitz D, et al. Increased titers of neutralizing antibodies after immunization with both envelope proteins of the porcine endogenous retroviruses (PERVs)[J]. Virol J, 2012, 9:260. DOI: 10.1186/1743-422X-9-260. [32] Waechter A, Denner J. Novel neutralising antibodies targeting the N-terminal helical region of the transmembrane envelope protein p15E of the porcine endogenous retrovirus (PERV)[J]. Immunol Res, 2014, 58(1):9-19. DOI: 10.1007/s12026-013-8430-y. [33] Langhammer S, Hübner J, Jarrett O, et al. Immunization with the transmembrane protein of a retrovirus, feline leukemia virus:absence of antigenemia following challenge[J]. Antiviral Res, 2011, 89(1):119-123. DOI: 10.1016/j.antiviral.2010.11.011. -





 下载:
下载: