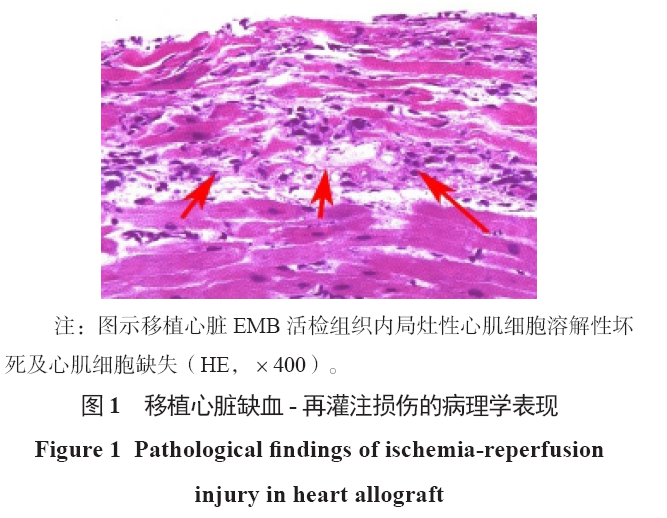
-
摘要: 随着心脏移植外科技术的提高和强效免疫抑制剂的临床应用,心脏移植例数和移植心脏存活时间均显著提升。然而在心脏移植术后的不同阶段,仍然可出现移植心脏右心室衰竭、缺血- 再灌注损伤、急性排斥反应、“Quilty病变”、感染和以移植心脏冠状动脉血管病(TCAD)为特征的慢性排斥反应等一系列并发症。心内膜心肌活组织检查(EMB)技术的应用使得包括排斥反应在内的移植心脏多种并发症的病理学特征得以观察和掌握,并成为移植心脏并发症最为准确的诊断手段。本文对移植心脏病理学研究的简史、心脏移植术后主要并发症及其诊断标准以及移植心脏排斥反应诊断研究的最新进展进行阐述,旨在使更多的心脏移植受者受益。Abstract: With the improvement of surgical technique of heart transplantation and clinical application of potent immunosuppressant, the quantity of heart transplantation and the survival time of heart allograft have been significantly improved. However, a series of complications, such as right ventricular failure, ischemia-reperfusion injury, acute rejection, "Quilty lesion", infection and chronic rejection characterized by transplant coronary artery disease (TCAD) may still occur at different stages after heart transplantation. The application of endomyocardial biopsy (EMB) makes it possible to observe and understand the pathological features of multiple complications of heart allograft including rejection, which has become the most accurate diagnostic tool for postoperative complications. In this article, the brief history of heart allograft pathology, main postoperative complications and pathological diagnostic criteria, and cutting edge research progress on diagnostic criteria of rejection were illustrated, aiming to bring clinical benefits to more recipients undergoing heart transplantation.
-
表 1 移植心脏AMR的病理学诊断分级(2013年ISHLT标准)
Table 1. Pathological diagnosis classification of AMR of heart allograft (2013 ISHLT working formulation)
分级 定义 病理学特征 pAMR0 病理学上无aAMR表现 组织病理学和免疫病理学染色均无表现 pAMR1(H+) 仅有单纯的病理学aAMR表现 具有组织病理学的表现,但C4d和(或)CD68免疫组化染色呈阴性 pAMR1(I+) 仅有单纯的免疫病理学aAMR表现 无组织病理学的表现,但C4d和(或)CD68免疫组化染色呈阳性 pAMR2 中度病理学的aAMR 同时具备了组织病理学和免疫组化染色阳性的表现 pAMR3 重度病理学的AMR 心肌间质出血及混合的炎症细胞浸润,血管内皮细胞水肿、核固缩和(或)核碎裂等损伤表现,心肌间质水肿并有免疫组化染色阳性;与重度的心脏血流动力学异常和移植心脏预后不良密切相关 -
[1] BILLINGHAM ME, CARY NR, HAMMOND ME, et al. A working formulation for the standardization of nomenclature in the diagnosis of heart and lung rejection: Heart Rejection Study Group. The International Society for Heart Transplantation[J]. J Heart Transplant, 1990, 9(6): 587-593. [2] BERRY GJ, BURKE MM, ANDERSEN C, et al. The 2013 International Society for Heart and Lung Transplantation Working Formulation for the standardization of nomenclature in the pathologic diagnosis of antibodymediated rejection in heart transplantation[J]. J Heart Lung Transplant, 2013, 32(12): 1147-1162. DOI: 10.1016/j.healun.2013.08.011. [3] DUONG VAN HUYEN JP, FEDRIGO M, FISHBEIN GA, et al. The XVth Banff Conference on Allograft Pathology the Banff Workshop Heart Report: improving the diagnostic yield from endomyocardial biopsies and Quilty effect revisited[J]. Am J Transplant, 2020, 20(12): 3308-3318. DOI: 10.1111/ajt.16083. [4] LOUPY A, HAAS M, ROUFOSSE C, et al. The Banff 2019 Kidney Meeting Report (I): updates on and clarification of criteria for T cell- and antibody-mediated rejection[J]. Am J Transplant, 2020, 20(9): 2318-2331. DOI: 10.1111/ajt.15898. [5] 黄洁, 杨跃进, 杨伟宪, 等. 心内膜心肌活检监测移植心脏排斥反应—附213例次心内膜活检结果分析[J]. 中华器官移植杂志, 2007, 28(11): 672-674. DOI: 10.3760/cma.j.issn.0254-1785.2007.11.009.HUANG J, YANG YJ, YANG WX, et al. Role of endomyocardial biopsy in rejection surveillance after heart transplantation: an analysis of 213 endomyocardial biopsies[J]. Chin J Organ Transplant, 2007, 28(11): 672-674. DOI: 10.3760/cma.j.issn.0254-1785.2007.11.009. [6] LI L, DUAN XJ, WANG HY, et al. Acute cellular rejection and antibody-mediated rejection in endomyocardial biopsy after heart transplantation: a retrospective study from a single medical center[J]. Int J Clin Exp Pathol, 2017, 10(4): 4772-4779. [7] STEWART S, WINTERS GL, FISHBEIN MC, et al. Revision of the 1990 working formulation for the standardization of nomenclature in the diagnosis of heart rejection[J]. J Heart Lung Transplant, 2005, 24(11): 1710- 1720. DOI: 10.1016/j.healun.2005.03.019. [8] CRESPO-LEIRO MG, VEIGA-BARREIRO A, DOMÉNECH N, et al. Humoral heart rejection (severe allograft dysfunction with no signs of cellular rejection or ischemia): incidence, management, and the value of C4d for diagnosis[J]. Am J Transplant, 2005, 5(10): 2560- 2564. DOI: 10.1111/j.1600-6143.2005.01039.x. [9] BRUNEVAL P, ANGELINI A, MILLER D, et al. The XⅢth Banff Conference on Allograft Pathology: the Banff 2015 Heart Meeting Report: improving antibodymediated rejection diagnostics: strengths, unmet needs, and future directions[J]. Am J Transplant, 2017, 17(1): 42- 53. DOI: 10.1111/ajt.14112. [10] SHETTY M, CHOWDHURY YS. Heart transplantation allograft vasculopathy[M]. Treasure Island (FL): StatPearls Publishing, 2021. [11] LOUPY A, TOQUET C, ROUVIER P, et al. Late failing heart allografts: pathology of cardiac allograft vasculopathy and association with antibody-mediated rejection[J]. Am J Transplant, 2016, 16(1): 111-120. DOI: 10.1111/ajt.13529. [12] SZYMANSKA S, GRAJKOWSKA W, SOBIESZCZANSKAMALEK M, et al. Prevalence of the Quilty effect in endomyocardial biopsy of patients after heart transplantation - from cellular rejection to antibody-mediated rejection?[J]. Pol J Pathol, 2016, 67(3): 216-220. DOI: 10.5114/pjp.2016.63772. [13] CHO H, CHOI JO, JEON ES, et al. Quilty lesions in the endomyocardial biopsies after heart transplantation[J]. J Pathol Transl Med, 2019, 53(1): 50-56. DOI: 10.4132/jptm.2018.11.30. [14] NOVÁK J, MACHÁČKOVÁ T, KREJČÍ J, et al. MicroRNAs as theranostic markers in cardiac allograft transplantation: from murine models to clinical practice[J]. Theranostics, 2021, 11(12): 6058-6073. DOI: 10.7150/thno.56327. [15] CONSTANSO-CONDE I, HERMIDA-PRIETO M, BARGE-CABALLERO E, et al. Circulating miR- 181a-5p as a new biomarker for acute cellular rejection in heart transplantation[J]. J Heart Lung Transplant, 2020, 39(10): 1100-1108. DOI: 10.1016/j.healun.2020.05.018. [16] GRANKVIST R, CHIREH A, SANDELL M, et al. Myocardial micro-biopsy procedure for molecular characterization with increased precision and reduced trauma[J]. Sci Rep, 2020, 10(1): 8029. DOI: 10.1038/s41598-020-64900-w. [17] NOVÁKOVÁ T, MACHÁČKOVÁ T, NOVÁK J, et al. Identification of a diagnostic set of endomyocardial biopsy microRNAs for acute cellular rejection diagnostics in patients after heart transplantation using nextgeneration sequencing[J]. Cells, 2019, 8(11): 1400. DOI: 10.3390/cells8111400. [18] ESCHER F, PIETSCH H, ALESHCHEVA G, et al. Evaluation of myocardial gene expression profiling for superior diagnosis of idiopathic giant-cell myocarditis and clinical feasibility in a large cohort of patients with acute cardiac decompensation[J]. J Clin Med, 2020, 9(9): 2689. DOI: 10.3390/jcm9092689. [19] LIN-WANG HT, CIPULLO R, DIAS FRANÇA JI, et al. Intragraft vasculitis and gene expression analysis: association with acute rejection and prediction of mortality in long-term heart transplantation[J]. Clin Transplant, 2018, 32(10): e13373. DOI: 10.1111/ctr.13373. [20] KHUSH KK, PATEL J, PINNEY S, et al. Noninvasive detection of graft injury after heart transplant using donor-derived cell-free DNA: a prospective multicenter study[J]. Am J Transplant, 2019, 19(10): 2889-2899. DOI: 10.1111/ajt.15339. [21] GIARRAPUTO A, BARISON I, FEDRIGO M, et al. A changing paradigm in heart transplantation: an integrative approach for invasive and non-invasive allograft rejection monitoring[J]. Biomolecules, 2021, 11(2): 201. DOI: 10.3390/biom11020201. [22] DENG MC. The evolution of patient-specific precision biomarkers to guide personalized heart-transplant care[J]. Expert Rev Precis Med Drug Dev, 2021, 6(1): 51-63. DOI: 10.1080/23808993.2021.1840273. [23] DENG MC, EISEN HJ, MEHRA MR, et al. Noninvasive discrimination of rejection in cardiac allograft recipients using gene expression profiling[J]. Am J Transplant, 2006, 6(1): 150-160. DOI: 10.1111/j.1600-6143.2005.01175.x. [24] NORTH PE, ZIEGLER E, MAHNKE DK, et al. Cellfree DNA donor fraction analysis in pediatric and adult heart transplant patients by multiplexed allelespecific quantitative PCR: validation of a rapid and highly sensitive clinical test for stratification of rejection probability[J]. PLoS One, 2020, 15(1): e0227385. DOI: 10.1371/journal.pone.0227385. [25] FITZSIMONS SJ, EVANS JDW, RASSL DM, et al. Highsensitivity cardiac troponin is not associated with acute cellular rejection after heart transplantation[J]. Transplantation, 2021, DOI: 10.1097/TP.0000000000003876[Epubaheadofprint]. [26] ERBEL C, TASKIN R, DOESCH A, et al. High-sensitive Troponin T measurements early after heart transplantation predict short- and long-term survival[J]. Transpl Int, 2013, 26(3): 267-272. DOI: 10.1111/tri.12024. [27] VACCHI E, BURRELLO J, DI SILVESTRE D, et al. Immune profiling of plasma-derived extracellular vesicles identifies Parkinson disease[J]. Neurol Neuroimmunol Neuroinflamm, 2020, 7(6): e866. DOI: 10.1212/NXI.0000000000000866. [28] BURRELLO J, BOLIS S, BALBI C, et al. An extracellular vesicle epitope profile is associated with acute myocardial infarction[J]. J Cell Mol Med, 2020, 24(17): 9945-9957. DOI: 10.1111/jcmm.15594. [29] LOUPY A, AUBERT O, ORANDI BJ, et al. Prediction system for risk of allograft loss in patients receiving kidney transplants: international derivation and validation study[J]. BMJ, 2019, 366: l4923. DOI: 10.1136/bmj.l4923. -





 下载:
下载: