-
摘要:
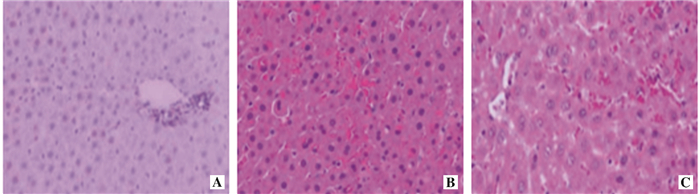
目的 探讨百里醌对肝缺血-再灌注损伤(IRI)的作用及其机制。 方法 30只C57小鼠随机均分为假手术(Sham)组、IRI组和百里醌(Thy)组(每组各10只)。术前1 h, Thy组给予百里醌(40 ml/kg)腹腔注射, Sham组和IRI组给予无水乙醇(40 ml/kg)腹腔注射。IRI组和Thy组建立小鼠肝IRI模型。再灌注4 h后收集血清和肝脏标本。光学显微镜下观察肝组织病理学改变, 并予病理损伤评分; 采用逆转录聚合酶链反应(RT-PCR)检测肝组织肿瘤坏死因子(TNF)-α、单核细胞趋化蛋白(MCP)-1和白细胞介素(IL)-6的信使核糖核酸(mRNA)表达水平; 采用酶链免疫吸附试验(ELISA)检测血清中TNF-α、MCP-1和IL-6的蛋白表达水平; 采用硫代巴比妥酸(TBA)法检测肝组织丙二醛(MDA)的含量; 采用ELISA法测定肝组织过氧化氢酶(CAT)、谷胱甘肽过氧化物酶(GPx)、超氧化物歧化酶(SOD)活性; 采用肝组织免疫印迹法(Western blot)检测Wnt、β-catenin、p53的蛋白表达水平。 结果 与Sham组比较, IRI组肝组织损伤严重, 损伤评分明显增加(P < 0.05), 肝组织和血清中的TNF-α、MCP-1、IL-6和肝组织MDA、Wnt、β-catenin、p53的表达均明显增多(P < 0.05~0.001), 而肝组织CAT、GPx与SOD活性均明显降低(均为P < 0.001)。与IRI组比较, Thy组肝组织损伤较轻, 损伤评分明显减少(P < 0.05), 肝组织和血清中的TNF-α、MCP-1、IL-6和肝组织MDA、Wnt、β-catenin、p53的表达均明显减少(均为P < 0.05), 而肝组织CAT、GPx与SOD活性均明显增高(均为P < 0.05)。 结论 百里醌通过减轻炎症反应和氧化应激而减轻肝IRI, 其作用机制与抑制Wnt/β-catenin/p53信号通路激活有关。 Abstract:Objective To investigate the effect and mechanism of thymoquinone on hepatic ischemia-reperfusion injury (IRI). Methods Thirty C57 mice were randomly divided into the sham operation (sham), IRI and thymoquinone (Thy) groups (n=10 in each group).At preoperative 1 h, thymoquinone at a dose of 40 ml/kg was administered via intraperitoneal injection in the Thy group. Absolute ethyl alcohol at the same dosage was given via intraperitoneal injection in the sham and IRI groups. Liver IRI mouse models were established in the IRI and Thy groups. Serum and liver specimens were collected at 4 h after reperfusion. Under light microscope, hepatic histopathological changes were observed and assessed by pathological injury grading. Reverse transcription polymerase chain reaction(RT-PCR) was performed to measure the messenger ribonucleic acid(mRNA) expression levels of tumor necrosis factor (TNF)-α, monocyte chemotactic protein (MCP)-1 and interleukin (IL)-6. The expression of TNF-α, MCP-1 and IL-6 proteins in the serum in the serum were assessed by ELISA. The content of malondialdehyde (MDA) in the liver tissue was detected by thiobarbituric acid (TBA). The activity of catalase (CAT), glutathioneperoxidase (GPx) and superoxide dismutase (SOD) in the liver tissue was determined by ELISA. The expression levels of Wnt, β-catenin and p53 proteins were measured by Western blot. Results Compared with the sham group, the liver injury was more severe and the hepatic injury grading was significantly enhanced in the IRI group (P < 0.05), the expression of TNF-α, MCP-1 and IL-6 in the liver tissue and serum sample, and MDA, Wnt, β-catenin and p53 in the liver tissue was significantly up-regulated (P < 0.05-0.001), whereas CAT, GPx and SOD activity in the liver tissue was dramatically reduced (all in P < 0.001). Compared with the IRI group, the liver injury in the Thy group was slighter and the liver injury grading was significantly decreased (both in P < 0.05). The expression levels of TNF-α, MCP-1 and IL-6 in the liver tissue and serum sample, MDA, Wnt, β-catenin and p53 in the liver tissue were significantly down-regulated (all in P < 0.05), whereas CAT, GPx and SOD activity was considerably up-regulated in the liver tissue (all in P < 0.05). Conclusions Thymoquinone can mitigate liver IRI through alleviating inflammatory response and oxidation stress. The underlying mechanism is correlated with inhibiting the activation of Wnt/β-catenin/p53 signaling pathway. -
Key words:
- Thymoquinone /
- Liver transplantation /
- Ischemia-reperfusion injury /
- Inflammation /
- Oxidation stress /
- Wnt /
- β-catenin /
- p53
-
表 1 本实验所用基因的引物序列
Table 1. Primer sequences of genes in the experiment
基因 种属 上游引物序列(5’ to 3’) 下游引物序列(3’ to 5’) TNF-α 小鼠 TACCCATACGATGTTCCAGATTACGCT TATCCATATGATGTTCCAGATTATGCT MCP-1 小鼠 TAATACGACTCACTATAGGG TAGAAGGCACAGTCGAGG IL-6 小鼠 GAGGATACCACTCCCAACAGACC AAGTGCATCATCGTTGTTCATACA β-actin 小鼠 GCTCTGGCTCCTAGCACCAT GCCACCGATCCACACAGAGT 表 2 3组小鼠肝组织中促炎症细胞因子mRNA表达水平的比较
Table 2. Comparison of mRNA levels of pro-inflammatory cytokines in the liver tissue of mice in three groups (x±s)
组别 n TNF-α mRNA MCP-1 mRNA IL-6 mRNA Sham组 10 1.00±0.10 1.00±0.05 0.85±0.10 IRI组 10 13.00±1.50b 5.50±0.40a 8.50±0.75a Thy组 10 5.00±0.50c 1.50±0.50c 2.50±0.50c 注:与Sham组比较,aP < 0.05,bP < 0.001;与IRI组比较,cP < 0.05 表 3 3组小鼠血清中促炎症细胞因子蛋白表达水平的比较
Table 3. Comparison of protein expression levels of pro-inflammatory cytokines in serum of mice in three groups(x±s, ng/L)
组别 n TNF-α MCP-1 IL-6 Sham组 10 110±10 100±20 100±10 IRI组 10 1 500±300b 650±40a 850±50a Thy组 10 450±150c 250±30c 550±30c 注:与Sham组比较,aP < 0.05,bP < 0.001;与IRI组比较,cP < 0.05 表 4 3组小鼠肝组织中氧化应激酶类活性的比较
Table 4. Comparison of oxidative stress enzymes activity in liver tissue of mice in three groups (x±s, ng/L)
组别 n CAT GPx SOD Sham组 10 100±10 95±5 103±9 IRI组 10 30±5a 40±4a 38±4a Thy组 10 60±5b 65±5b 75±7b 注:与Sham组比较,aP < 0.001;与IRI组比较,bP < 0.05 -
[1] Patel RP, Lang JD, Smith AB, et al. Redox therapeutics in hepatic ischemia reperfusion injury[J]. World J Hepatol, 2014, 6(1):1-8. http://www.cqvip.com/qk/71422x/201401/1003031655.html [2] Datta G, Fuller BJ, Davidson BR. Molecular mechanisms of liver ischemia reperfusion injury:insights from transgenic knockout models[J]. World J Gastroenterol, 2013, 19(11):1683-1698. doi: 10.3748/wjg.v19.i11.1683 [3] Dalal AR. Split liver transplantation:what's unique[J]. World J Transplant, 2015, 5(3):89-94. doi: 10.5500/wjt.v5.i3.89 [4] Gracia-Sancho J, Casillas-Ramírez A, Peralta C. Molecular pathways in protecting the liver from ischaemia/reperfusion injury:a 2015 update[J].Clin Sci, 2015, 129(4):345-362. doi: 10.1042/CS20150223 [5] Weeder PD, van Rijn R, Porte RJ. Machine perfusion in liver transplantation as a tool to prevent non-anastomotic biliary strictures:rationale, current evidence and future directions[J]. J Hepatol, 2015, 63(1):265-275. doi: 10.1016/j.jhep.2015.03.008 [6] Darakhshan S, Pour AB, Colagar AH, et al. Thymoquinone and its therapeutic potentials[J]. Pharmacol Res, 2015, 95/96:138-158. doi: 10.1016/j.phrs.2015.03.011 [7] Ahmad A, Husain A, Mujeeb M, et al. A review on therapeutic potential of Nigella sativa:a miracle herb[J]. Asian Pac J Trop Biomed, 2013, 3(5):337-352. doi: 10.1016/S2221-1691(13)60075-1 [8] Majdalawieh AF, Fayyad MW. Immunomodulatory and anti-inflammatory action of Nigella sativa and thymoquinone:a comprehensive review[J].Int Immunopharmacol, 2015, 28(1):295-304. doi: 10.1016/j.intimp.2015.06.023 [9] Khader M, Eckl PM. Thymoquinone:an emerging natural drug with a wide range of medical applications[J]. Iran J Basic Med Sci, 2014, 17(12):950-957. http://europepmc.org/articles/PMC4387230 [10] Tao X, Sun X, Yin L, et al. Dioscin ameliorates cerebral ischemia/reperfusion injury through the downregulation of TLR4 signaling via HMGB-1 inhibition[J]. Free Radic Biol Med, 2015, 84:103-115. doi: 10.1016/j.freeradbiomed.2015.03.003 [11] Liu A, Huang L, Fan H, et al. Baicalein pretreatment protects against liver ischemia/reperfusion injury via inhibition of NF-κB pathway in mice[J]. Int Immunopharmacol, 2015, 24(1):72-79. doi: 10.1016/j.intimp.2014.11.014 [12] Mcdonald KA, Huang H, Tohme S, et al. Toll-like receptor 4(TLR4) antagonist eritoran tetrasodium attenuates liver ischemia and reperfusion injury through inhibition of high-mobility group box protein B1(HMGB1) signaling[J]. Mol Med, 2015, 20:639-648. http://molmed.org/journal/articles/37/1736 [13] Guan LY, Fu PY, Li PD, et al. Mechanisms of hepatic ischemia-reperfusion injury and protective effects of nitric oxide[J]. World J Gastrointest Surg, 2014, 6(7):122-128. doi: 10.4240/wjgs.v6.i7.122 [14] 李夏静, 陈瑞琦, 陈旭征, 等.总丹参多酚酸预处理对脊髓缺血再灌注损伤的保护作用[J/CD].中华细胞与干细胞杂志(电子版), 2015, 5(2):33-37. http://www.cnki.com.cn/Article/CJFDTotal-ZXGA201502008.htmLi XJ, Chen RQ, Chen XZ, et al. Protective effect of pretreatment with salvianolic acids on spinal cord ischemia-reperfusion injury in rats[J/CD]. Chin J Cell and Stem Cell(Electr Edit), 2015, 5(2):33-37. http://www.cnki.com.cn/Article/CJFDTotal-ZXGA201502008.htm [15] 马帅军, 张更, 曹志强, 等.肾缺血-再灌注损伤大鼠SDF-1、ICAM-1表达与肾小管坏死评分的相关性研究[J].器官移植, 2014, 5(5):294-298. http://www.organtranspl.com/browse/detail/qkid/82/id/192.htmlMa SJ, Zhang G, Cao ZQ, et al. Relationship between the expression of SDF-1, ICAM-1 and renal tubular necrosis score in rats with renal ischemic reperfusion injury[J]. Organ Transplant, 2014, 5(5):294-298. http://www.organtranspl.com/browse/detail/qkid/82/id/192.html [16] Chtourou Y, Aouey B, Kebieche, et al. Protective role of naringin against cisplatin induced oxidative stress, inflammatory response and apoptosis in rat striatum via suppressing ROS-mediated NF-κB and P53 signaling pathways[J]. Chem Biol Interact, 2015, 239:76-86. doi: 10.1016/j.cbi.2015.06.036 [17] Zhao L, An R, Yang Y, et al. Melatonin alleviates brain injury in mice subjected to cecal ligation and puncture via attenuating inflammation, apoptosis, and oxidative stress:the role of SIRT1 signaling[J]. J Pineal Res, 2015, 59(2):230-239. doi: 10.1111/jpi.12254 [18] Zhang F, Ren T, Wu J, et al. Small concentrations of TGF-β1 promote proliferation of bone marrow-derived mesenchymal stem cells via activation of Wnt/β-catenin pathway[J]. Indian J Exp Biol, 2015, 53(8):508-513. http://www.researchgate.net/publication/281636862_Small_concentrations_of_TGF-1_promote_proliferation_of_bone_marrow-derived_mesenchymal_stem_cells_via_activation_of_Wnt-catenin_pathway [19] Li XL, Zhou J, Chen ZR, et al. P53 mutations in colorectal cancer:molecular pathogenesis and pharmacological reactivation[J]. World J Gastroenterol, 2015, 21(1):84-93. doi: 10.3748/wjg.v21.i1.84 [20] Dibra D, Mishra L, Li S. Molecular mechanisms of oncogene-induced inflammation and inflammation-sustained oncogene activation in gastrointestinal tumors:an under-appreciated symbiotic relationship[J]. Biochim Biophys Acta, 2014, 1846(1):152-160. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4140981/ -





 下载:
下载: