Effect of nodosin on accelerating hepatocyte regeneration after partial liver transplantation in rats
-
摘要:
目的 探讨溪黄草有效成分nodosin对大鼠减体积肝移植术后肝细胞再生的影响。 方法 以Wistar大鼠作为供体, SD大鼠作为受体, 采用改良的二袖套法建立大鼠部分肝移植模型, 将24只受体大鼠随机分为nodosin组和对照组。nodosin组于肝移植术后经尾静脉注射nodosin 100μg/ml。在术后3 d和7 d分别处死大鼠取外周血浆及肝脏组织标本。分光光度法检测外周血浆中丙氨酸转氨酶(ALT)、天冬氨酸转氨酶(AST)和白蛋白(ALB)含量; 光学显微镜下观察肝组织形态学改变; 采用免疫组织化学法检测肝组织增殖细胞核抗原(PCNA)抗体的表达水平; 采用蛋白印迹法(Western blot)检测肝组织中磷酸化蛋白激酶(p-AKT)、磷酸化哺乳动物雷帕霉素靶蛋白(p-mTOR)、细胞周期素D1(cyclin D1)、血红素加氧酶(HO)-1蛋白的表达水平; 采用磷脂酰丝氨酸外翻分析(Annexin V法)和dUTP缺口末端标记(TUNEL)法检测肝细胞的凋亡情况。 结果 术后3 d和7 d, 与对照组比较, nodosin组大鼠血清中ALT、AST含量较低, ALB含量较高(均为P < 0.05), 同时nodosin能够减轻大鼠肝移植术后肝组织病理损伤。nodosin组的PCNA阳性表达细胞数明显多于对照组(P < 0.05)。nodosin组增殖相关蛋白p-AKT、p-mTOR、cyclin D1和HO-1蛋白表达水平均明显高于对照组(均为P < 0.05)。nodosin组发生凋亡的肝细胞数量和比例明显低于对照组(P < 0.05)。 结论 大鼠肝移植术后应用nodosin具有减少肝细胞凋亡数量, 促进肝细胞增殖的作用。 Abstract:Objective To evaluate the effect of nodosin, as an effective element extracted from rabdosiae serrae, on hepatocyte regeneration after partial liver transplantation. Methods Wistar rats were used as donors and SD rats as recipients. Rat models with partial liver transplantation were established by modified two-cuff technique. Twenty-four recipient rats were randomly assigned into the nodosin and control groups. In the nodosin group, nodosin at a dosage of 100 μg/ml was administered via tail venous route after liver transplantation. Peripheral plasma and liver specimen were obtained at postoperative 3 and 7 d. The levels of alanine transaminase (ALT), aspartate aminotransferase (ALT) and albumin (ALB) in the peripheral plasma were measured by spectrophotometry. Hepatic histomorphological changes were observed under light microscope. The positive cell count of proliferating cell nuclear antigen (PCNA) antibody in the liver tissue was detected by immunohistochemistry. The expression levels of phosphorylated protein kinase (p-AKT), phosphorylated mammalian target of rapamycin (p-mTOR), cyclin D1 and heme oxygenase (HO)-1 proteins were measured by western blot. The apoptosis of liver cells was detected by Annexin V method and TdT mediated-dUTP nick end labeling (TUNEL). Results Compared with the control group, the serum levels of ALT and AST were significantly lower at 3 d and 7 d after operation, whereas the ALB content was significantly higher in the nodosin group (all in P < 0.05). And nodosin could alleviate the pathological injury of rat liver tissue after transplantation. The positive cell count of PCNA in the nodosin group was significantly higher than that in the control group (P < 0.05). In the nodosin group, the expression levels of p-AKT, p-mTOR, cyclin D1and HO-1 proteins were significantly higher than those in the control group(all in P < 0.05). The quantity and percentage of apoptotic hepatocytes in the nodosin group were significantly lower than those in the control group (both in P < 0.05). Conclusions Application of nodosin can decrease the quantity of apoptotic hepatocytes and accelerate hepatocyte proliferation after liver transplantation in rat models. -
Key words:
- Nodosin /
- Liver regeneration /
- Liver transplantation /
- Hepatocyte, apoptosis /
- Hepatocyte, proliferation
-
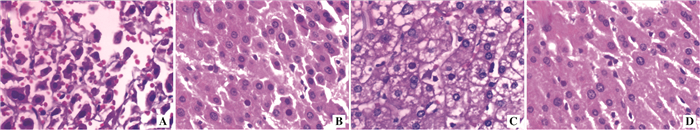
图 2 两组大鼠术后3 d和7 d肝细胞PCNA阳性细胞数的比较
注:A图为对照组术后3 d肝细胞PCNA的免疫组化染色切片(DAB,×100);B图为nodosin组术后3 d肝细胞PCNA的免疫组化染色切片(DAB,×100);C图为对照组术后7 d肝细胞PCNA的免疫组化染色切片(DAB,×100);D为nodosin组术后7 d肝细胞PCNA的免疫组化染色切片(DAB,×100);E图为两组PCNA蛋白阳性表达数量对比,与对照组相比,aP < 0.05
Figure 2. Comparison of positive cell count of PCNA in hepatocyte of rats in two groups at 3 d and 7 d after operation
图 4 两组大鼠术后3 d和7 d肝细胞的凋亡情况
注:A图为对照组术后3 d流式细胞图;B图为nodosin组术后3 d流式细胞图;C图为对照组术后7 d流式细胞图;D图为nodosin组术后7 d流式细胞图;E图为两组大鼠肝细胞凋亡比例的比较,与对照组相比,aP < 0.05;F图为对照组术后3 d肝细胞凋亡情况(TUNEL,×100);G图为nodosin组术后3 d肝细胞凋亡情况(TUNEL,×100);H图为对照组术后7 d肝细胞凋亡情况(TUNEL,×100);I图为nodosin组术后7 d肝细胞凋亡情况(TUNEL,×100);J图为两组大鼠肝细胞凋亡指数的比较,与对照组相比,aP < 0.05
Figure 4. Apoptosis of hepatocyte of rats in two groups at 3 d and 7 d after operation
表 1 两组大鼠术后3 d和7 d血清中ALT、AST和ALB的含量
Table 1. Contents of ALT, AST and ALB in serum of rats in two groups at 3 d and 7 d after operation (x±s)
组别 n ALT(mmol/L) AST(mmol/L) ALB(g/L) 术后3 d 术后7 d 术后3 d 术后7 d 术后3 d 术后7 d nodosin组 12 136±8a 86±9a 167±8a 100±9a 19.7±1.9a 23.6±1.4a 对照组 12 226±12 135±6 281±10 160±21 17.4±1.6 19.1±0.7 注:与对照组比较,aP < 0.05 -
[1] Lee SG. A complete treatment of adult living donor liver transplantation: a review of surgical technique and current challenges to expand indication of patients[J]. Am J Transplant, 2015, 15(1):17-38. doi: 10.1111/ajt.12907 [2] Wan P, Yu X, Xia Q. Operative outcomes of adult living donor liver transplantation and deceased donor liver transplantation: a systematic review and Meta-analysis[J]. Liver Transpl, 2014, 20(4):425-436. doi: 10.1002/lt.v20.4 [3] 杜隽铭, 吴萍, 孙丽娟, 等.溪黄草有效成分nodosin灌注对大鼠移植肝脏病理损伤的影响[J].器官移植, 2011, 2(1):9-13. http://www.organtranspl.com/browse/detail/qkid/33/id/445.htmlDu JM, Wu P, Sun LJ, et al. Effect of nodosin perfusion on rat liver gratis pathological injury[J]. Organ Transplant, 2011, 2(1):9-13. http://www.organtranspl.com/browse/detail/qkid/33/id/445.html [4] 王健东, 杜隽铭, 李济宇, 等. HO-1诱导剂体外灌注对大鼠移植肝脏保护作用的研究[J].器官移植, 2010, 1(2):103-106. http://www.cnki.com.cn/Article/CJFDTotal-QGYZ201002012.htmWang JD, Du JM, Li JY, et al. Protection of liver grafts with HO-1 inducer by extra-corporeal liver perfusion in rats[J]. Organ Transplant, 2010, 1(2):103-106. http://www.cnki.com.cn/Article/CJFDTotal-QGYZ201002012.htm [5] Li J, Du J, Sun L, et al. Anti-inflammatory function of nodosin via inhibition of IL-2[J]. Am J Chin Med, 2010, 38(1):127-142. doi: 10.1142/S0192415X10007713 [6] Wang H, Li C, Hu J, et al. Effect of different suprahepatic vena cava reconstruction methods on the hemodynamics of rats after liver transplantation[J]. PLoS One, 2013, 8(9):e72695. doi: 10.1371/journal.pone.0072695 [7] Olthoff KM, Emond JC, Shearon TH, et al. Liver regeneration after living donor transplantation: adult-to-adult living donor liver transplantation cohort study[J]. Liver Transpl, 2015, 21(1):79-88. doi: 10.1002/lt.v21.1 [8] Fujiyoshi M, Ozaki M. Molecular mechanisms of liver regeneration and protection for treatment of liver dysfunction and diseases[J]. J Hepatobiliary Pancreat Sci, 2011, 18(1):13-22. doi: 10.1007/s00534-010-0304-2 [9] Lin L, Zhuang M, Zou L, et al. Structural characteristics of water-soluble polysaccharides from Rabdosia serra (MAXIM.) HARA leaf and stem and their antioxidant capacities[J]. Food Chem, 2012, 135(2):730-737. doi: 10.1016/j.foodchem.2012.05.029 [10] Ryter SW, Choi AM. Targeting heme oxygenase-1 and carbon monoxide for therapeutic modulation of inflammation[J]. Transl Res, 2016, 167(1):7-34. doi: 10.1016/j.trsl.2015.06.011 [11] He XH, Tang JJ, Wang YL, et al. Transduced heme oxygenase-1 fusion protein reduces renal ischemia/reperfusion injury through its antioxidant and antiapoptotic roles in rats[J]. Transplant Proc, 2015, 47(6):1627-1632. doi: 10.1016/j.transproceed.2015.04.098 [12] Wei J, Fan G, Zhao H, et al. Heme oxygenase-1 attenuates inflammation and oxidative damage in a rat model of smoke-induced emphysema[J]. Int J Mol Med, 2015, 36(5):1384-1392. [13] Unek G, Ozmen A, Mendilcioglu I, et al. The expression of cell cycle related proteins PCNA, Ki67, p27 and p57 in normal and preeclamptic human placentas[J]. Tissue Cell, 2014, 46(3):198-205. doi: 10.1016/j.tice.2014.04.003 [14] Unek G, Ozmen A, Ozekinci M, et al. Immunolocalization of cell cycle proteins (p57, p27, cyclin D3, PCNA and Ki67) in intrauterine growth retardation (IUGR) and normal human term placentas[J]. Acta Histochem, 2014, 116(3):493-502. doi: 10.1016/j.acthis.2013.10.007 [15] Sozmen M, Devrim AK, Tunca R, et al. Protective effects of silymarin on fumonisin B1-induced hepatotoxicity in mice[J]. J Vet Sci, 2014, 15(1):51-60. doi: 10.4142/jvs.2014.15.1.51 [16] Paplomata E, O'Regan R. The PI3K/AKT/mTOR pathway in breast cancer: targets, trials and biomarkers[J]. Ther Adv Med Oncol, 2014, 6(4):154-166. doi: 10.1177/1758834014530023 [17] Chen J, Alberts I, Li X. Dysregulation of the IGF-I/PI3K/AKT/mTOR signaling pathway in autism spectrum disorders[J]. Int J Dev Neurosci, 2014, 35:35-41. doi: 10.1016/j.ijdevneu.2014.03.006 [18] 蒋超, 石厚霞, 丁思加, 等. mTOR信号通路在iPS定向分化RPE细胞中的调控机制研究[J/CD].中华细胞与干细胞杂志(电子版), 2014, 4(1):36-43.Jiang C, Shi HX, Ding SJ, et al. The mechanism of mTOR signaling pathway in the regulation of differentiation of iPS into retinal pigment epithelial cells[J/CD]. Chin J Cell Stem Cell(Electr Edit), 2014, 4(1):36-43. [19] 张艳, 李志樑, 胡春玲, 等. mTOR/p70S6K信号通路在冠心病患者外周血T细胞中的表达及活化[J].实用医学杂志, 2014, 30(21):3437-3440. http://www.cnki.com.cn/Article/CJFDTOTAL-SYYZ201421026.htmZhang Y, Li ZL, Hu CL, et al. Study on the activation of mTOR/p70S6K signaling pathway in peripheral T lymphocytes of the CHD patients[J]. Pract Med J, 2014, 30(21):3437-3440. http://www.cnki.com.cn/Article/CJFDTOTAL-SYYZ201421026.htm [20] Zhou Q, Liu C, Liu W, et al. Rotenone induction of hydrogen peroxide inhibits mTOR-mediated S6K1 and 4E-BP1/eIF4E pathways, leading to neuronal apoptosis[J]. Toxicol Sci, 2015, 143(1):81-96. doi: 10.1093/toxsci/kfu211 [21] Xu Y, Liu C, Chen S, et al. Activation of AMPK and inactivation of Akt result in suppression of mTOR-mediated S6K1 and 4E-BP1 pathways leading to neuronal cell death in in vitro models of Parkinson's disease[J]. Cell Signal, 2014, 26(8):1680-1689. doi: 10.1016/j.cellsig.2014.04.009 -





 下载:
下载: