Research report of living donor kidney harvesting in Bama miniature pigs with six gene modified
-
摘要:
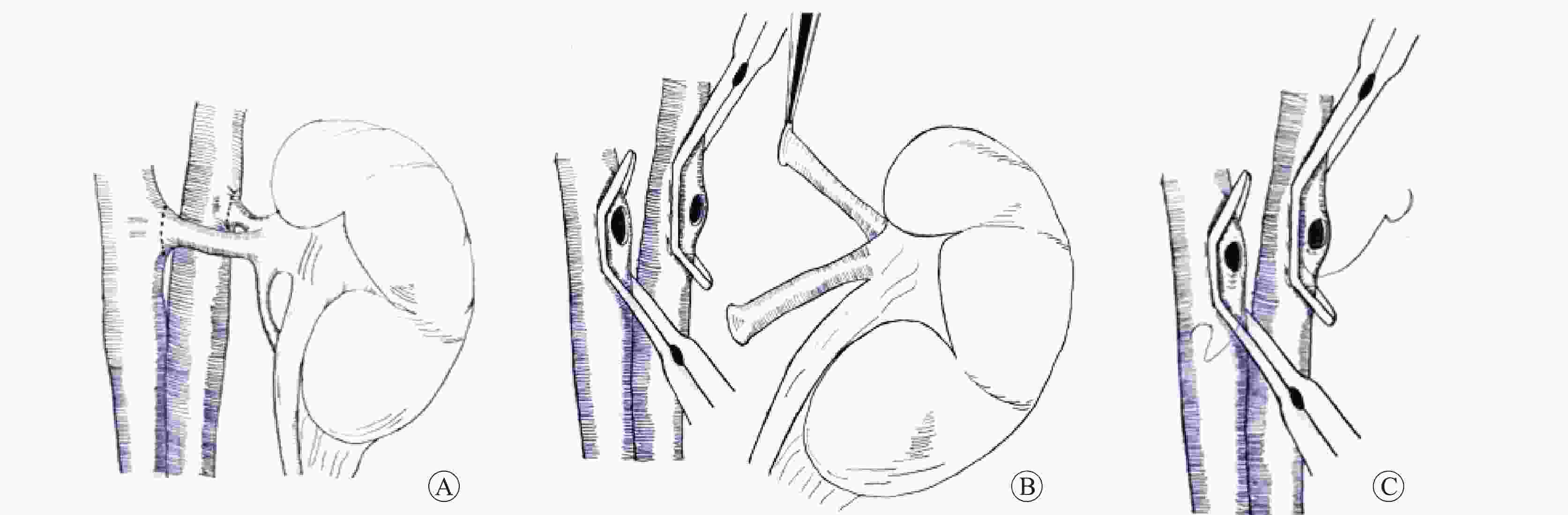
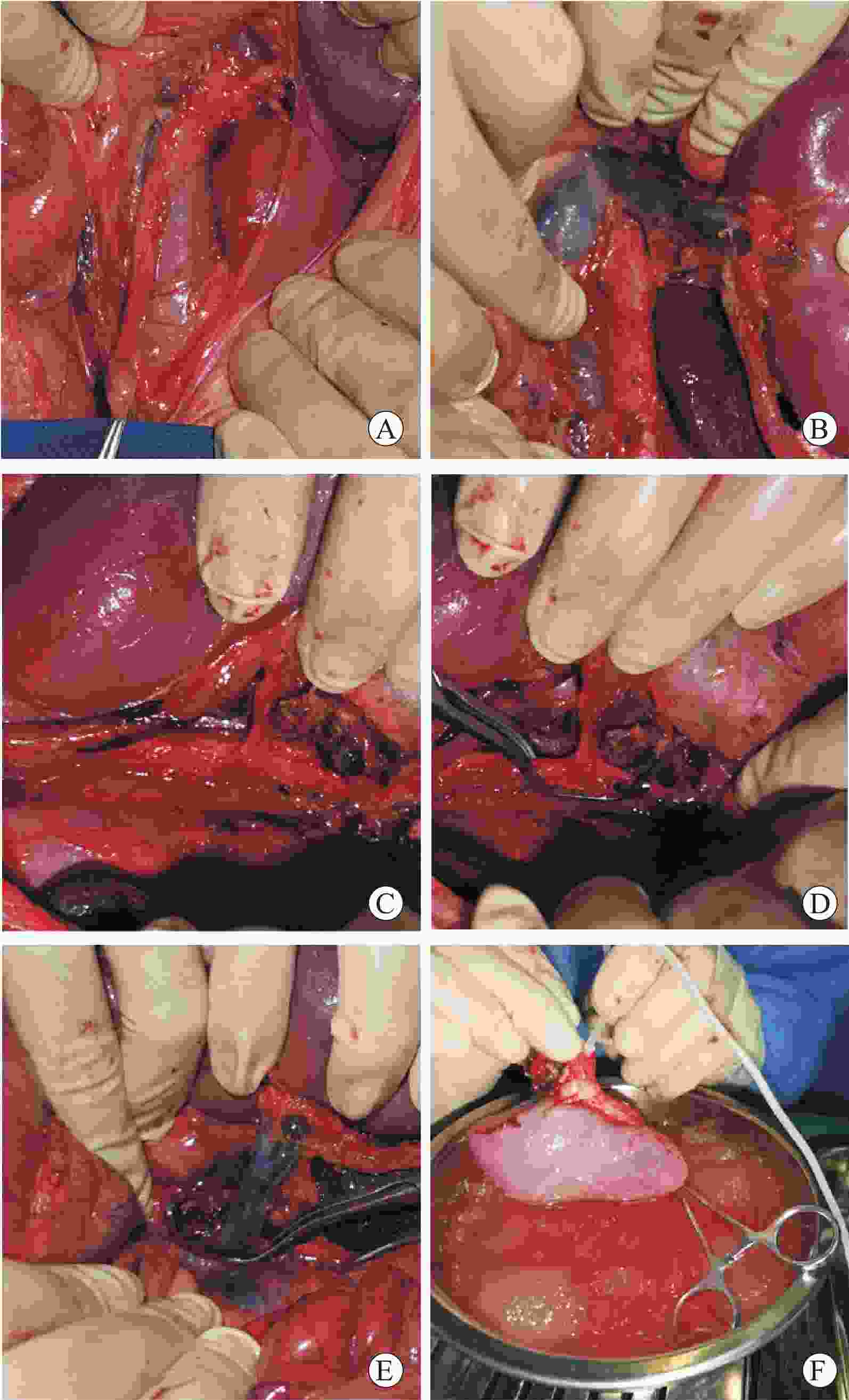
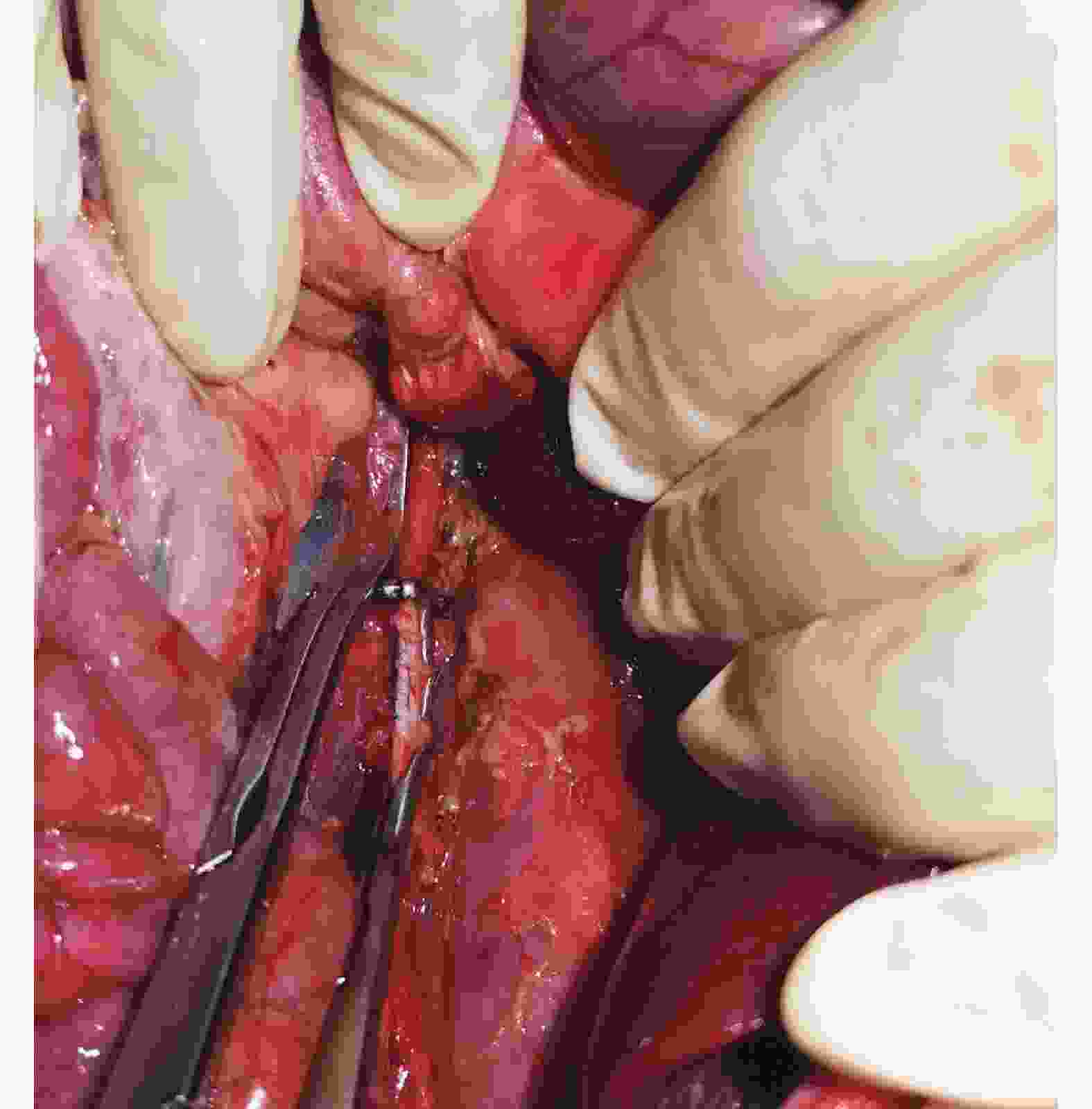
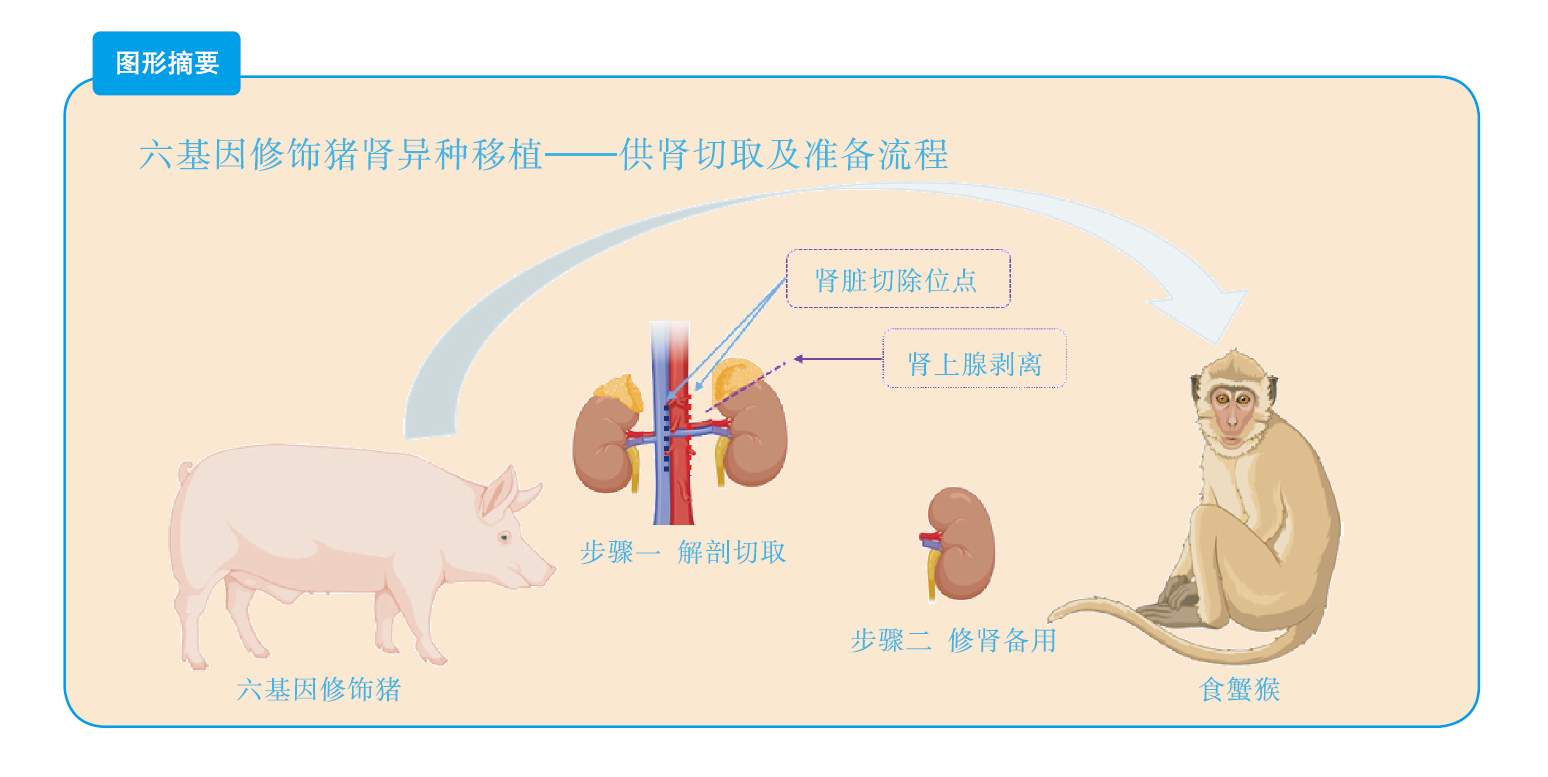
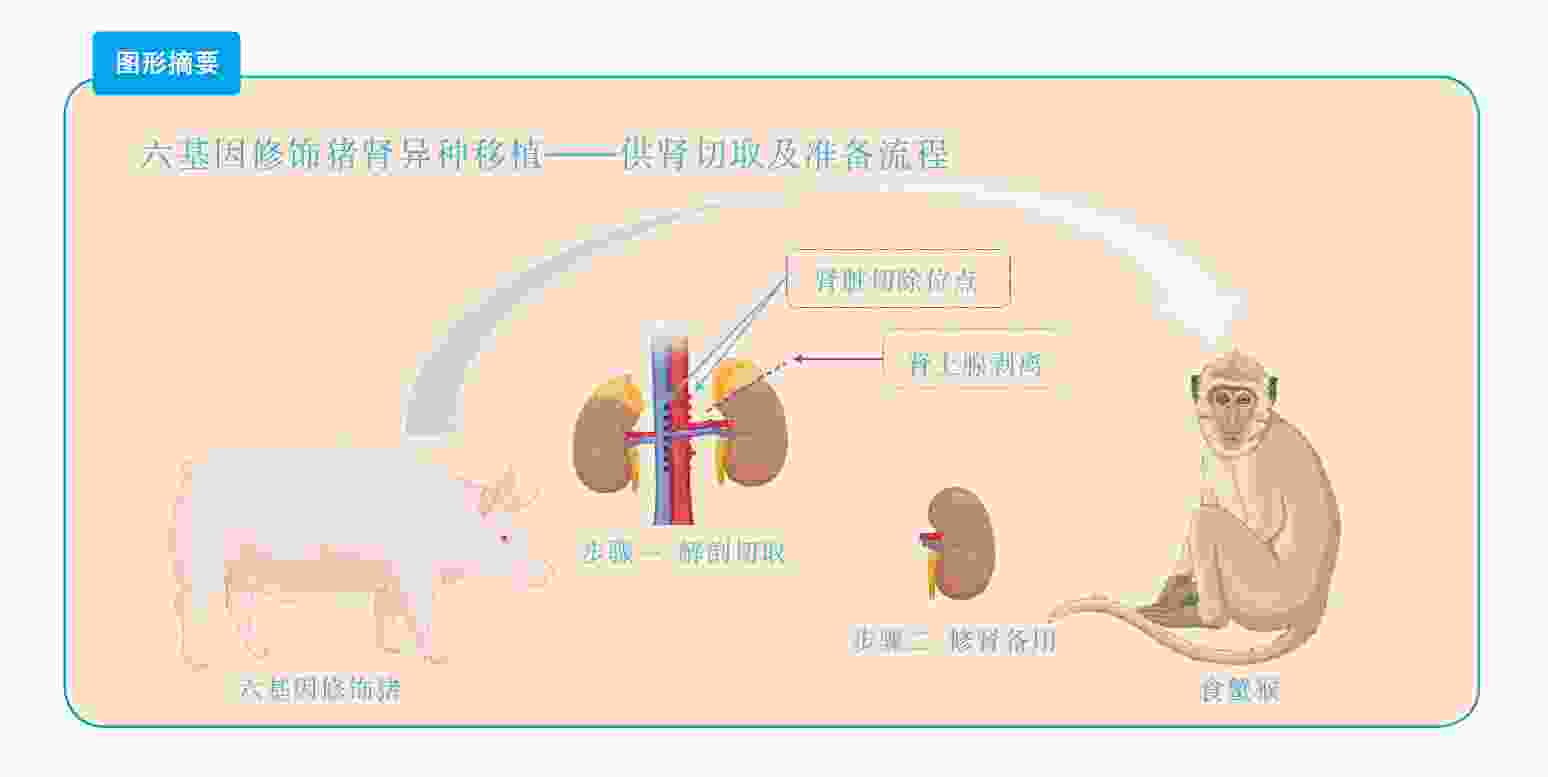
目的 总结基因修饰猪活体供肾切取的经验及其实践价值。 方法 采用活体供肾切取技术,切取六基因修饰猪左侧肾脏。首先阻断输尿管,游离下腔静脉及腹主动脉后,切取过程中依次完成输尿管、肾静脉、肾动脉的显露、游离。在腹主动脉、下腔静脉上阻断钳,离断肾动脉、静脉后立即用4 ℃肾保存液进行灌注,保存在冰生理盐水中准备移植。同时完成供体腹主动脉、下腔静脉缺口的缝合。记录手术时间,出血量,热缺血和冷缺血时间,并发症发生情况以及供、受体存活情况。 结果 成功切取基因修饰猪左肾,取肾术中出血5 mL,热缺血时间45 s,冷缺血时间2.5 h。供、受体均未输血,植入受体内的肾脏恢复泌尿功能。切取左肾后供体健康存活8个月余。 结论 基因修饰猪活体供肾切取技术安全可靠,游离肾脏过程中同时处理分支血管,减少了修肾过程,缩短了冷缺血时间,活体供肾切取有助于后续供体存活并进行其他科学研究。 Abstract:Objective To summarize the experience and practical value of living donor kidney harvesting in Bama miniature pigs with six gene modified. Methods The left kidney of Bama miniature pigs with six gene modified was obtained by living donor kidney harvesting technique. First, the ureter was occluded, and then the inferior vena cava and abdominal aorta were freed. During the harvesting process, the ureter, renal vein and renal artery were exposed and freed in sequence. The vascular forceps were used at the abdominal aorta and inferior vena cava, and the renal artery and vein were immediately perfused with 4℃ renal preservation solution, and stored in ice normal saline for subsequent transplantation. Simultaneously, the donor abdominal aorta and inferior vena cava gap were sutured. The operation time, blood loss, warm and cold ischemia time, postoperative complications and the survival of donors and recipients were recorded. Results The left kidney of the genetically modified pig was successfully harvested. Intraoperative bleeding was 5 mL, warm ischemia time was 45 s, and cold ischemia time was 2.5 h. Neither donor nor recipient pig received blood transfusion, and urinary function of the kidney transplanted into the recipient was recovered. The donor survived for more than 8 months after the left kidney was resected. Conclusions Living donor kidney harvesting is safe and reliable in genetically modified pigs. Branch blood vessels could be processed during kidney harvesting, which shortens the process of kidney repair and the time of cold ischemia. Living donor kidney harvesting contributes to subsequent survival of donors and other scientific researches. -
表 1 供体猪血常规检测
Table 1. Routine blood test of the donor pig
时间 红细胞
(×1012/L)白细胞
(×109/L)淋巴细胞
(×109/L)血红蛋白
(g/L)红细胞比容 平均红细胞
体积(/fL)平均红细胞血红
蛋白量(/pg)平均红细胞血红
蛋白浓度(g/L)术前 6.02 11.21 7.56 120 0.34 54.5 18.3 344 术后8个月 5.83 11.79 6.87 116 0.31 59.1 20.6 342 正常值 5.50~9.00 10.20~30.00 4.80~16.20 100~160 0.33~0.52 51.0~73.0 14.0~22.0 300~360 -
[1] SYKES M, SACHS DH. Transplanting organs from pigs to humans[J]. Sci Immunol, 2019, 4(41): eaau6298. DOI: 10.1126/sciimmunol.aau6298. [2] YAMADA K, ARIYOSHI Y, POMPOSELLI T, et al. Co-transplantation of vascularized thymic graft with kidney in pig-to-nonhuman primates for the induction of tolerance across xenogeneic barriers[J]. Methods Mol Biol, 2020, 2110: 151-171. DOI: 10.1007/978-1-0716-0255-3_11. [3] 赵恒, 徐凯祥, 范柠粼, 等. 基因编辑异种移植供体猪的构建及现状[J/CD]. 实用器官移植电子杂志, 2018, 6(5): 412-418. DOI: 10.3969/j.issn.2095-5332.2018.05.018.ZHAO H, XU KX, FAN NL, et al. Construction and current status of genetically edited xenograft donor pigs [J/CD]. Pract J Organ Transplant(Electr Vers), 2018, 6(5): 412-418. DOI: 10.3969/j.issn.2095-5332.2018.05.018. [4] VERNOOIJ RWM, LAW W, PETERS SAE, et al. The probability of receiving a kidney transplantation in end-stage kidney disease patients who are treated with haemodiafiltration or haemodialysis: a pooled individual participant data from four randomised controlled trials[J]. BMC Nephrol, 2021, 22(1): 70. DOI: 10.1186/s12882-021-02265-6. [5] BURGAN CM, SUMMERLIN D, LOCKHART ME. Renal transplantation: pretransplant workup, surgical techniques, and surgical anatomy[J]. Radiol Clin North Am, 2023, 61(5): 797-808. DOI: 10.1016/j.rcl.2023.04.003. [6] WANG Y, LEI T, WEI L, et al. Xenotransplantation in China: present status[J]. Xenotransplantation, 2019, 26(1): e12490. DOI: 10.1111/xen.12490. [7] COZZI E, SCHNEEBERGER S, BELLINI MI, et al. Organ transplants of the future: planning for innovations including xenotransplantation[J]. Transpl Int, 2021, 34(11): 2006-2018. DOI: 10.1111/tri.14031. [8] COOPER DKC, HARA H, IWASE H, et al. Pig kidney xenotransplantation: progress toward clinical trials[J]. Clin Transplant, 2021, 35(1): e14139. DOI: 10.1111/ctr.14139. [9] RODGER D, HURST DJ, COOPER DK. Xenotransplantation: a historical-ethical account of viewpoints[J]. Xenotransplantation, 2023, 30(2): e12797. DOI: 10.1111/xen.12797. [10] SYKES M, SACHS DH. Progress in xenotransplantation: overcoming immune barriers[J]. Nat Rev Nephrol, 2022, 18(12): 745-761. DOI: 10.1038/s41581-022-00624-6. [11] ARABI TZ, SABBAH BN, LERMAN A, et al. Xenotransplantation: current challenges and emerging solutions[J]. Cell Transplant, 2023, 32: 9636897221148771. DOI: 10.1177/09636897221148771. [12] ANAND RP, LAYER JV, HEJA D, et al. Design and testing of a humanized porcine donor for xenotransplantation[J]. Nature, 2023, 622(7982): 393-401. DOI: 10.1038/s41586-023-06594-4. [13] SINGH AK, CHAN JL, SEAVEY CN, et al. CD4+CD25Hi FoxP3+ regulatory T cells in long-term cardiac xenotransplantation[J]. Xenotransplantation, 2018, 25(2): e12379. DOI: 10.1111/xen.12379. [14] HAWTHORNE WJ. World first pig-to-human cardiac xenotransplantation[J]. Xenotransplantation, 2022, 29(1): e12733. DOI: 10.1111/xen.12733. [15] MOHIUDDIN MM, SINGH AK, SCOBIE L, et al. Graft dysfunction in compassionate use of genetically engineered pig-to-human cardiac xenotransplantation: a case report[J]. Lancet, 2023, 402(10399): 397-410. DOI: 10.1016/S0140-6736(23)00775-4. [16] BALIGA RR. Genetically modified porcine-to-human cardiac xenotransplantation[J]. N Engl J Med, 2022, 387(14): 1337-1338. DOI: 10.1056/NEJMc2210401. [17] MOAZAMI N, STERN JM, KHALIL K, et al. Pig-to-human heart xenotransplantation in two recently deceased human recipients[J]. Nat Med, 2023, 29(8): 1989-1997. DOI: 10.1038/s41591-023-02471-9. [18] PORRETT PM, ORANDI BJ, KUMAR V, et al. First clinical-grade porcine kidney xenotransplant using a human decedent model[J]. Am J Transplant, 2022, 22(4): 1037-1053. DOI: 10.1111/ajt.16930. [19] MONTGOMERY RA, STERN JM, LONZE BE, et al. Results of two cases of pig-to-human kidney xenotransplantation[J]. N Engl J Med, 2022, 386(20): 1889-1898. DOI: 10.1056/NEJMoa2120238. [20] LOUPY A, GOUTAUDIER V, GIARRAPUTO A, et al. Immune response after pig-to-human kidney xenotransplantation: a multimodal phenotyping study[J]. Lancet, 2023, 402(10408): 1158-1169. DOI: 10.1016/S0140-6736(23)01349-1. [21] NIH National Library of Medicine. Porcine kidney xenotransplantation in patients with end-stage kidney disease[EB/OL]. [2023-10-01]. https://clinicaltrials.gov/study/NCT05340426. [22] LEE K, FARRELL K, UH K. Application of genome-editing systems to enhance available pig resources for agriculture and biomedicine[J]. Reprod Fertil Dev, 2019, 32(2): 40-49. DOI: 10.1071/RD19273. [23] PETERSEN B, FRENZEL A, LUCAS-HAHN A, et al. Efficient production of biallelic GGTA1 knockout pigs by cytoplasmic microinjection of CRISPR/Cas9 into zygotes[J]. Xenotransplantation, 2016, 23(5): 338-346. DOI: 10.1111/xen.12258. [24] COOPER DKC. What will be the cost of a genetically-engineered pig organ for clinical xenotransplantation?[J]. Xenotransplantation, 2020, 27(5): e12606. DOI: 10.1111/xen.12606. [25] 刘圣圳, 董隽, 罗光达, 等. 机器人辅助腹腔镜活体供肾切取术(附2例报告)[J]. 微创泌尿外科杂志, 2015(2): 75-77. DOI: 10.3969/j.issn.2095-5146.2015.02.003.LIU SZ, DONG J, LUO GD, et al. Robotic-assisted living donor nephrectomy (two cases of reports)[J]. J Minim Invasive Urol, 2015(2): 75-77. DOI: 10.3969/j.issn.2095-5146.2015.02.003. [26] 董隽, 卢锦山, 祖强, 等. 改良手辅助后腹腔镜活体供肾切取术(附视频)[J/CD]. 中华移植杂志(电子版), 2010, 4(3): 219-220. DOI: 10.3877/cma.j.issn.1674-3903.2010.03.010.DONG J, LU JS, ZU Q, et al. Modified hand-assisted retrolaparoscopic living donor nephrectomy [J/CD]. Chin J Transplant (Electr Edit), 2010, 4(3): 219-220. DOI: 10.3877/cma.j.issn.1674-3903.2010.03.010. [27] OZTURK SA, YUKSEL Y, ERBIS H, et al. Laparoscopic live donor nephrectomy: experience of high-volume center with 2, 477 cases[J]. Urol Int, 2021, 105(1/2): 100-107. DOI: 10.1159/000511377. [28] TIONG HY, GOH BYS, CHIONG E, et al. Robotic kidney autotransplantation in a porcine model: a procedure-specific training platform for the simulation of robotic intracorporeal vascular anastomosis[J]. J Robot Surg, 2018, 12(4): 693-698. DOI: 10.1007/s11701-018-0806-5. [29] 王西墨. 猪到猕猴异种肾移植的实验研究[D]. 湖北: 华中科技大学, 2001. DOI: 10.7666/d.y392889. [30] 郭霜, 李选鹏, 满江位, 等. 手助后腹腔镜和开放活体供肾切取术的临床分析[J]. 微创泌尿外科杂志, 2019, 8(1): 1-6. DOI: 10.19558/j.cnki.10-1020/r.2019.01.001.GUO S, LI XP, MAN JW, et al. Clinical analysis of hand-assisted retroperitoneoscopic and open living donor nephrectomy[J]. J Minim Invasive Urol, 2019, 8(1): 1-6. DOI: 10.19558/j.cnki.10-1020/r.2019.01.001. [31] 雷永生, 王志伟, 王钢, 等. 后腹腔镜右侧活体供肾切取术单中心经验[J/CD]. 中华移植杂志(电子版), 2020, 14(4): 234-236. DOI: 10.3877/cma.j.issn.1674-3903.2020.04.008.LEI YS, WANG ZW, WANG G, et al. Single center experience in retrolaparoscopic right living donor nephrectomy [J/CD] . Chin J Transplant (Electr Edit), 2020, 14(4): 234-236. DOI: 10.3877/cma.j.issn.1674-3903.2020.04.008. [32] DAGNÆS-HANSEN J, KRISTENSEN GH, STROOMBERG HV, et al. Surgical approaches and outcomes in living donor nephrectomy: a systematic review and meta-analysis[J]. Eur Urol Focus, 2022, 8(6): 1795-1801. DOI: 10.1016/j.euf.2022.03.021. [33] WINDISCH OL, MATTER M, PASCUAL M, et al. Robotic versus hand-assisted laparoscopic living donor nephrectomy: comparison of two minimally invasive techniques in kidney transplantation[J]. J Robot Surg, 2022, 16(6): 1471-1481. DOI: 10.1007/s11701-022-01393-x. [34] NEWMAN ME, MUSK GC, HE B. Establishment of laparoscopic live donor nephrectomy in a porcine model: techniques and outcomes in 44 pigs[J]. J Surg Res, 2018, 222: 132-138. DOI: 10.1016/j.jss.2017.09.042. [35] GKEKA K, TSATURYAN A, FAITATZIADIS S, et al. Robot-assisted radical nephrectomy using the novel avatera robotic surgical system: a feasibility study in a porcine model[J]. J Endourol, 2023, 37(3): 273-278. DOI: 10.1089/end.2022.0596. [36] PELEGRIN T, CHAMPY CM, GERBAUD F, et al. Robotic-assisted laparoscopy living donor nephrectomy: technique and results of a monocentric retrospective series[J]. Prog Urol, 2022, 32(8/9): 567-576. DOI: 10.1016/j.purol.2022.03.009. [37] KOGA S, YAMANAGA S, HIDAKA Y, et al. Influence of graft ureter length, a donor-related factor, on urinary tract infections after living-donor kidney transplantation: a single-center analysis of 211 cases[J]. Transpl Int, 2022, 35: 10754. DOI: 10.3389/ti.2022.10754. [38] FOOTE JB, BIKHET MH, HANSEN-ESTRUCH C, et al. Observations on hydronephrosis after pig kidney transplantation in baboons[J]. Xenotransplantation, 2022, 29(6): e12779. DOI: 10.1111/xen.12779. -




 下载:
下载: