Application of calcineurin inhibitor monotherapy in renal transplantation after alemtuzumab induction: a Meta-analysis
-
摘要:
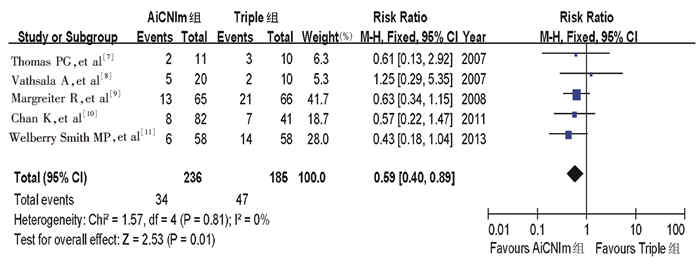
目的 探讨阿仑单抗诱导后钙神经蛋白抑制剂单药治疗(AiCNIm)方案应用于肾移植术后免疫抑制的有效性和安全性。 方法 应用计算机检索Pubmed、Embase、Web of science、Cochrance library及中国知网(CNKI)等数据库, 查找1980年至2014年12月31日发表的有关AiCNIm方案(AiCNIm组)和常规三联方案(Triple组)进行肾移植术后免疫抑制的随机对照临床研究。采用Rev Man 5.2软件进行Meta分析。 结果 共纳入5项随机对照研究, 421例肾移植受者。随访6~12个月结果显示, 与Triple组相比, AiCNIm组的急性排斥反应或穿刺活检证实的排斥反应发生率更低[相对风险系数(RR)=0.59, 95%可信区间(CI)0.40~0.89], 但两组在移植肾失功(RR=0.85, 95%CI0.38~1.87)、受者死亡(RR=0.89, 95%CI0.30~2.67)、感染(RR=1.03, 95%CI0.91~1.17)和移植后新发糖尿病(RR=0.62, 95%CI0.29~1.30)方面比较, 差异无统计学意义(均为P>0.05)。 结论 基于现有证据, 肾移植术后应用阿仑单抗诱导后用钙神经蛋白抑制剂单药治疗, 短期免疫抑制效果好且安全。 Abstract:Objective To evaluate the clinical efficacy and safety of immunosuppression of calcineurin inhibitor monotherapy (AiCNIm) after alemtuzumab induction following renal transplantation. Methods Randomized control clinical trials related to application of AiCNIm (AiCNIm group) and conventional triple regimes (Triple group) for immunosupression after renal transplantation, published from 1980 to December 31 2014, were searched online from PubMed, Embase, Web of Science, Cochrance library and China National Knowledge Infrastructure (CNKI)databases. Meta-analysis was performed by Rev Man 5.2 software. Results Five randomized control studies consisting of 421 renal transplant recipients were included. The results of follow up for 6-12 months revealed that compared with the Triple group, the incidence of rejection response confirmed by acute rejection or aspiration biopsy in the AiCNIm group was significantly lower [relative risk(RR)=0.59, 95% confidence interval (CI): 0.40-0.89]. However, there was no significant difference in the risk of renal allograft dysfunction (RR=0.85, 95%CI: 0.38-1.87), death of recipient (RR=0.89, 95%CI: 0.30-2.67), infection (RR=1.03, 95%CI: 0.91-1.17) and new-onset diabetes after transplantation(RR=0.62, 95%CI: 0.29-1.30) between two groups(all in P>0.05). Conclusions According to the existing evidence, application of calcineurin inhibitor monotherapy after renal transplantation exerts short-term immunosuppressive effect and high safety after alemtuzumab induction. -
Key words:
- Renal transplantation /
- Alemtuzumab /
- Calcineurin inhibitor /
- Randomized control test /
- Meta-analysis
-
表 1 纳入文献的基本特征
Table 1. Basic characteristics of included literature
研究作者 发表年份 AiCNIm组 Triple组 随访时间 供者类型 免疫危险度(PRA抗体) n 诱导剂量 CNI n 诱导方案 三联方案 Thomas PG, et al[7] 2007 11 Alem(30 mg,1次) FK506 10 rATG FK506+MMF+Pred 127~611 d DD 高危 Vathsala A, et al[8] 2007 20 Alem(20 mg,2次) CsA 10 None CsA+AZA+Pred 6个月 LD, DD 低危 Margreiter R, et al[9] 2008 65 Alem(20 mg,2次) FK506 66 None FK506+MMF+Pred 12个月 DD 低危 Chan K, et al[10] 2011 82 Alem(30 mg,1次) FK506 41 Dac FK506+MMF+Pred 12个月 LD, DD 低危 Welberry Smith MP, et al[11] 2013 58 Alem(30 mg,1次) FK506 58 Bas FK506+MMF 12个月 DBD, DCD, LD 中、低危 注:Alem为阿仑单抗;FK506为他克莫司;CsA为环孢素;rATG为兔抗胸腺细胞球蛋白;Dac为达利珠单抗;Bas为巴利昔单抗;MMF为吗替麦考酚酯;Pred为泼尼松;AZA为硫唑嘌呤;DD为尸体供者;LD为活体供者;DBD为脑死亡器官捐献供者;DCD为心脏死亡器官捐献供者 -
[1] Mori M. Alemtuzumab, a monoclonal antibody against CD52: hopes and fears[J]. Brain Nerve, 2014, 66(10):1179-1189. http://www.ncbi.nlm.nih.gov/pubmed/25296872 [2] Coles AJ, Fox E, Vladic A, et al. Alemtuzumab versus interferon β-1a in early relapsing-remitting multiple sclerosis: post-hoc and subset analyses of clinical efficacy outcomes[J]. Lancet Neurol, 2011, 10(4):338-348. doi: 10.1016/S1474-4422(11)70020-5 [3] Dhaun N, Kluth DC. Alemtuzumab induction therapy in kidney transplantation[J]. Lancet, 2015, 385(9970):770. doi: 10.1016/S0140-6736(15)60431-7 [4] Tan HP, Kaczorowski DJ, Basu A, et al. Living donor renal transplantation using alemtuzumab induction and tacrolimus monotherapy[J]. Am J Transplant, 2006, 6(10):2409-2417. doi: 10.1111/ajt.2006.6.issue-10 [5] 刘银春, 葛龙, 李雅睿, 等. PRISMA声明在评价《循证医学》刊载的干预类系统评价/荟萃分析报告质量中的应用[J].中华医学图书情报杂志, 2014, 23(2): 24-27. http://www.cnki.com.cn/Article/CJFDTOTAL-YXTS201402007.htmLiu YC, Ge L, Li YR, et al. Application of PRISMA statement in assessment of intervention-related systematic reviews and meta-analyses published in evidence-based medicine[J]. Chin J Med Libr Inf Sci, 2014, 23(2): 24-27. http://www.cnki.com.cn/Article/CJFDTOTAL-YXTS201402007.htm [6] Mehrazmay A, Karambakhsh A, Salesi M. Reporting quality assessment of randomized controlled trials published in Nephrology Urology Monthly Journal[J]. Nephrourol Mon, 2015, 7(4): e28752. http://paper.medlive.cn/literature/1462384 [7] Thomas PG, Woodside KJ, Lappin JA, et al. Alemtuzumab (Campath 1H) induction with tacrolimus monotherapy is safe for high immunological risk renal transplantation[J]. Transplantation, 2007, 83(11):1509-1512. doi: 10.1097/01.tp.0000263344.53000.a1 [8] Vathsala A, Ona ET, Tan SY, et al. Randomized trial of alemtuzumab for prevention of graft rejection and preservation of renal function after kidney transplantation[J]. Transplantation, 2005, 80(6):765-774. doi: 10.1097/01.tp.0000166921.14670.33 [9] Margreiter R, Klempnauer J, Neuhaus P, et al. Alemtuzumab (Campath-1H) and tacrolimus monotherapy after renal transplantation: results of a prospective randomized trial[J]. Am J Transplant, 2008, 8(7):1480-1485. doi: 10.1111/j.1600-6143.2008.02273.x [10] Chan K, Taube D, Roufosse C, et al. Kidney transplantation with minimized maintenance: alemtuzumab induction with tacrolimus monotherapy:an open label, randomized trial[J]. Transplantation, 2011, 92(7):774-780. doi: 10.1097/TP.0b013e31822ca7ca [11] Welberry Smith MP, Cherukuri A, Newstead CG, et al. Alemtuzumab induction in renal transplantation permits safe steroid avoidance with tacrolimus monotherapy: a randomized controlled trial[J]. Transplantation, 2013, 96(12):1082-1088. doi: 10.1097/TP.0b013e3182a64db9 [12] Hanaway MJ, Woodle ES, Mulgaonkar S, et al. Alemtuzumab induction in renal transplantation[J]. N Engl J Med, 2011, 364(20):1909-1919. doi: 10.1056/NEJMoa1009546 [13] Shin M, Song SH, Kim JM, et al. Alemtuzumab induction in deceased donor kidney transplantation[J]. Transplant Proc, 2011, 43(6):2365-2378. doi: 10.1016/j.transproceed.2011.05.032 [14] Morgan RD, O'Callaghan JM, Knight SR, et al. Alemtuzumab induction therapy in kidney transplantation: a systematic review and Meta-analysis[J]. Transplantation, 2012, 93(12):1179-1188. doi: 10.1097/TP.0b013e318257ad41 [15] Cannon RM, Brock G, Marvin MR, et al. Analysis of BK viral infection after alemtuzumab induction for renal transplant[J]. Transpl Infect Dis, 2012, 14(4):374-379. doi: 10.1111/tid.2012.14.issue-4 [16] Helfrich M, Ison MG. Opportunistic infections complicating solid organ transplantation with alemtuzumab induction[J]. Transpl Infect Dis, 2015, 17(5):627-636. doi: 10.1111/tid.2015.17.issue-5 [17] Peleg AY, Husain S, Kwak EJ, et al. Opportunistic infections in 547 organ transplant recipients receiving alemtuzumab, a humanized monoclonal CD-52 antibody[J]. Clin Infect Dis, 2007, 44(2):204-212. doi: 10.1086/510388 [18] Ciancio G, Gaynor JJ, Guerra G, et al. Randomized trial of three induction antibodies in kidney transplantation: long-term results[J]. Transplantation, 2014, 97(11):1128-1138. doi: 10.1097/01.TP.0000441089.39840.66 [19] Sung J, Barry JM, Jenkins R, et al. Alemtuzumab induction with tacrolimus monotherapy in 25 pediatric renal transplant recipients[J]. Pediatr Transplant, 2013, 17(8):718-725. doi: 10.1111/petr.2013.17.issue-8 [20] Haynes R, Baigent C, Harden P, et al. Campath, calcineurin inhibitor reduction and chronic allograft nephropathy (3C) study: background, rationale, and study protocol[J]. Transplant Res, 2013, 2(1):7. doi: 10.1186/2047-1440-2-7 -





 下载:
下载: