Study on effects of curcumin pretreatment on intestinal ischemia-reperfusion injury
-
摘要:
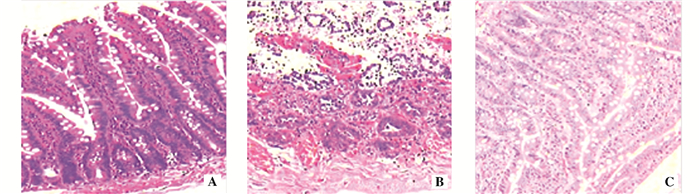
目的 探讨姜黄素预处理对大鼠肠缺血-再灌注损伤(IRI)的作用及其机制。 方法 将30只SD大鼠随机均分为假手术组(Sham组)、IRI组和姜黄素预处理组(Cur组),每组10只。Cur组术前1 h给予姜黄素腹腔注射(5 mg/kg),Sham组和IRI组则给予等体积生理盐水腹腔注射,IRI组和Cur组行小肠IRI手术。观察3组大鼠肠组织病理学变化。采用逆转录聚合酶链反应法检测肠组织白细胞介素(IL)-6、IL-8和肿瘤坏死因子(TNF)-α的信使RNA (mRNA)水平。采用酶链免疫吸附试验(ELISA)检测血清中IL-6、IL-8和TNF-α的蛋白表达水平。采用硫代巴比妥酸反应物法检测肠组织丙二醛(MDA)含量,采用ELISA检测肠组织过氧化氢酶(CAT)、谷胱甘肽过氧化物酶(GPx)和超氧化物歧化酶(SOD)含量。采用蛋白印迹法检测肠组织中磷脂酰肌醇-3-羟激酶(PI3K)、蛋白激酶(AKT)和哺乳动物雷帕霉素靶蛋白(mTOR)的蛋白表达水平。 结果 与Sham组相比,IRI组肠组织损伤程度,肠组织与血清中的IL-6、IL-8、TNF-α、MDA、PI3K、AKT、mTOR的表达水平均明显升高,而肠组织CAT、GPx与SOD含量明显降低。与IRI组相比,Cur组肠组织损伤程度,肠组织与血清中的IL-6、IL-8、TNF-α、MDA、PI3K、AKT、mTOR的表达水平均明显降低,而肠组织CAT、GPx与SOD含量明显升高(均为P<0.05)。 结论 姜黄素对大鼠发生IRI的肠组织有保护作用,该保护作用可能与抑制PI3K/AKT/mTOR信号通路介导的氧化应激和炎症有关。 Abstract:Objective To explore the effects and mechanism of curcumin pretreatment on intestinal ischemia-reperfusion injury (IRI) in rats. Methods Thirty SD rats were randomly divided into sham operation group (Sham group), IRI group and curcumin pretreatment group (Cur group), with 10 rats in each group. Intraperitoneal injection with curcumin (5 mg/kg) was given to rats in Cur group at 1 h before operation; intraperitoneal injection with normal saline of same volume was given to rats in Sham group and IRI group. Rats in IRI group and Cur group received intestinal IRI operation. Pathological changes in intestinal tissues of the rats in 3 groups were observed. Messenger RNA (mRNA) levels of interleukin (IL)-6, IL-8 and tumor necrosis factor (TNF)-α in intestinal tissue were detected by reverse transcription polymerase chain reaction, and protein expression levels of IL-6, IL-8 and TNF-α in serum were detected by enzyme linked immunosorbent assay (ELISA). Malondialdehyde(MDA) in intestinal tissue was detected by thiobarbituric acid reactive substance assay, as well as contents of catalase(CAT), glutathione peroxidase(GPx) and superoxide dismutase(SOD) in intestinal tissue were detected by ELISA. Protein expression levels of phosphatidylinositol 3-kinase (PI3K), protein kinase (AKT) and mammalian target of rapamycin (mTOR) in intestinal tissue were detected by Western blotting. Results Compared with Sham group, the injury degree of intestinal tissue as well as the expression levels of IL-6, IL-8, TNF-α, MDA, PI3K, AKT and mTOR in intestinal tissue and serum increased significantly, while the contents of CAT, GPx and SOD in intestinal tissue decreased significantly in IRI group. Compared with IRI group, the injury degree of intestinal tissue as well as the expression levels of IL-6, IL-8, TNF-α, MDA, PI3K, AKT and mTOR in intestinal tissue and serum decreased significantly, while the contents of CAT, GPx and SOD in intestinal tissue increased significantly in Cur group (all in P<0.05). Conclusions Curcumin presents protective effect on intestinal tissue with IRI in rats, which may be related to the inhibition of oxidative stress and inflammation mediated by PI3K/AKT/mTOR signaling pathway. -
Key words:
- Curcumin /
- Ischemia-reperfusion injury /
- Intestinal transplantation /
- Oxidative stress
-
表 1 本实验所用基因的引物序列
Table 1. Primer sequences of genes in the experiment
基 因 种属 上游引物序列(5’to 3’) 下游引物序列(3’to 5’) TNF-α 大鼠 CTGAACTTCGGGGTGATCGG GGCTTGTCACTCGAATTTGAGA IL-8 大鼠 CTGCAAGAGACTTCCATCCAG AGTGGTATAGACAGGTCTGTTG IL-6 大鼠 AGCTTCCTTGTGCAAGTGTC GACAGCCCAGGTCAAAGGTT β-actin 大鼠 AGAGGGAAATCGTGCGTGAC CAATAGTGATGACCTGGCCGT 表 2 3组大鼠肠组织IL-6、IL-8和TNF-α的mRNA表达水平的比较
Table 2. Comparison of mRNA levels of IL-6, IL-8 and TNF-α in the intestinal tissue of rats in three groups (x±s)
组 别 n IL-6 mRNA IL-8 mRNA TNF-α mRNA Sham组 10 1.0±0.1 1.0±0.2 1.0±0.1 IRI组 10 6.0±0.8b 5.0±0.4a 14.5±2.8a Cur组 10 2.0±0.3c 2.5±0.3c 6.5±0.8c 注:与Sham组比较,aP<0.05,bP<0.001;与IRI组比较,cP<0.05 表 3 3组大鼠血清中IL-6、IL-8和TNF-α蛋白表达水平的比较
Table 3. Comparison of protein expression levels of IL-6, IL-8 and TNF-α in serum of rats in three groups (x±s, ng/L)
组 别 n IL-6 IL-8 TNF-α Sham组 10 100±20 100±10 100±20 IRI组 10 900±100b 650±50a 1 700±150a Cur组 10 400±50c 350±40c 850±100c 注:与Sham组比较,aP<0.05,bP<0.001;与IRI组比较,cP<0.05 表 4 3组大鼠肠组织中MDA、CAT、GPx和SOD含量的比较
Table 4. Comparison of MDA、CAT、GPx and SOD content in intestinal tissue of rats in three groups (x±s, ng/L)
组 别 n MDA CAT GPx SOD Sham组 10 100±20 100±7 105±15 106±9 IRI组 10 1 700±300b 35±5a 32±4a 28±5a Cur组 10 500±150c 51±5c 60±5c 75±7c 注:与Sham组比较,aP<0.05,bP<0.001;与IRI组比较,cP<0.05 表 5 3组大鼠肠组织中PI3K、AKT和mTOR蛋白表达水平的比较
Table 5. Comparison of protein expression levels of PI3K, AKT and mTOR in intestinal tissue of rats in three groups (x±s)
组 别 n PI3K AKT mTOR Sham组 10 0.07±0.03 0.06±0.02 0.13±0.02 IRI组 10 0.31±0.03b 0.65±0.07a 0.55±0.05a Cur组 10 0.18±0.02d 0.29±0.04d 0.22±0.03c 注:与Sham组比较,aP<0.05,bP<0.01;与IRI组比较,cP<0.05,dP<0.01 -
[1] Gonzalez LM, Moeser AJ, Blikslager AT. Animal models of ischemia-reperfusion-induced intestinal injury: progress and promise for translational research[J]. Am J Physiol Gastrointest Liver Physiol,2015,308(2):G63-G75. doi: 10.1152/ajpgi.00112.2013 [2] Oltean M. Intestinal preservation for transplantation: current status and alternatives for the future[J]. Curr Opin Organ Transplant,2015,20(3):308-313. doi: 10.1097/MOT.0000000000000187 [3] Oltean M, Churchill TA. Organ-specific solutions and strategies for the intestinal preservation[J]. Int Rev Immunol,2014,33(3):234-244. doi: 10.3109/08830185.2013.853764 [4] Lenaerts K, Ceulemans LJ, Hundscheid IH, et al. New insights in intestinal ischemia-reperfusion injury: implications for intestinal transplantation[J]. Curr Opin Organ Transplant,2013,18(3):298-303. doi: 10.1097/MOT.0b013e32835ef1eb [5] Wong DM, Moore RM, Brockus CW. Intestinal ischemia-reperfusion injury in horses: pathogenesis and therapeutics[J]. Compend Contin Educ Vet,2012,34(8):E5. http://cn.bing.com/academic/profile?id=2399267034&encoded=0&v=paper_preview&mkt=zh-cn [6] Bhattacharyya A, Chattopadhyay R, Mitra S, et al. Oxidative stress: an essential factor in the pathogenesis of gastrointestinal mucosal diseases[J]. Physiol Rev,2014,94(2):329-354. doi: 10.1152/physrev.00040.2012 [7] Von Websky MW, Kalff JC, Schäfer N. Current knowledge on regulation and impairment of motility after intestinal transplantation[J]. Curr Opin Organ Transplant,2015,20(3):303-307. doi: 10.1097/MOT.0000000000000190 [8] Gibellini L, Bianchini E, De Biasi S, et al. Natural compounds modulating mitochondrial functions[J]. Evid Based Complement Alternat Med,2015:527209. http://cn.bing.com/academic/profile?id=1564135916&encoded=0&v=paper_preview&mkt=zh-cn [9] Fiorentini D, Zambonin L, Vieceli Dalla Sega F, et al. Polyphenols as modulators of aquaporin family in health and disease[J]. Oxid Med Cell Longev,2015:196914. http://cn.bing.com/academic/profile?id=1559856110&encoded=0&v=paper_preview&mkt=zh-cn [10] Mahmood K, Zia KM, Zuber M, et al. Recent developments in curcumin and curcumin based polymeric materials for biomedical applications: a review[J]. Int J Biol Macromol,2015,81:877-890. doi: 10.1016/j.ijbiomac.2015.09.026 [11] Ghosh S, Banerjee S, Sil PC. The beneficial role of curcumin on inflammation, diabetes and neurodegenerative disease: a recent update[J]. Food Chem Toxicol,2015,83:111-124. doi: 10.1016/j.fct.2015.05.022 [12] Shanmugam MK, Rane G, Kanchi MM, et al. The multifaceted role of curcumin in cancer prevention and treatment[J]. Molecules,2015,20(2):2728-2769. doi: 10.3390/molecules20022728 [13] Fan Z, Yao J, Li Y, et al. Anti-inflammatory and antioxidant effects of curcumin on acute lung injury in a rodent model of intestinal ischemia reperfusion by inhibiting the pathway of NF-Kb[J]. Int J Clin Exp Pathol,2015,8(4):3451-3459. http://cn.bing.com/academic/profile?id=2418151191&encoded=0&v=paper_preview&mkt=zh-cn [14] Arumugam TV, Okun E, Tang SC, et al. Toll-like receptors in ischemia-reperfusion injury[J]. Shock,2009,32(1):4-16. doi: 10.1097/SHK.0b013e318193e333 [15] Aivatidi C, Vourliotakis G, Georgopoulos S, et al. Oxidative stress during abdominal aortic aneurysm repair: biomarkers and antioxidant's protective effect: a review[J]. Eur Rev Med Pharmacol Sci, 2011,15(3):245-252. http://cn.bing.com/academic/profile?id=2098113086&encoded=0&v=paper_preview&mkt=zh-cn [16] 马帅军,张更,曹志强,等.肾缺血-再灌注损伤大鼠SDF-1、ICAM-1表达与肾小管坏死评分的相关性研究[J].器官移植,2014,5(5):294-298. http://www.organtranspl.com/browse/detail/qkid/82/id/192.htmlMa SJ, Zhang G, Cao ZQ, et al. Relationship between the expression of SDF-1,ICAM-1 and renal tubular necrosis score in rats with renal ischemic reperfusion injury[J]. Organ Transplant, 2014,5(5):294-298. http://www.organtranspl.com/browse/detail/qkid/82/id/192.html [17] Huang CY, Hsiao JK, Lu YZ, et al. Anti-apoptotic PI3K/Akt signaling by sodium/glucose transporter 1 reduces epithelial barrier damage and bacterial translocation in intestinal ischemia[J]. Lab Invest,2011,91(2):294-309. doi: 10.1038/labinvest.2010.177 [18] Hao F, Kang J, Cao Y, et al. Curcumin attenuates palmitate-induced apoptosis in MIN6 pancreatic β-cells through PI3K/Akt/FoxO1 andmitochondrial survival pathways[J]. Apoptosis,2015,20(11):1420-1432. doi: 10.1007/s10495-015-1150-0 -





 下载:
下载: