Efficacy analysis of T lymphocyte polyclonal antibody in renal transplantation from donor kidney of organ donation after citizen's death
-
摘要:
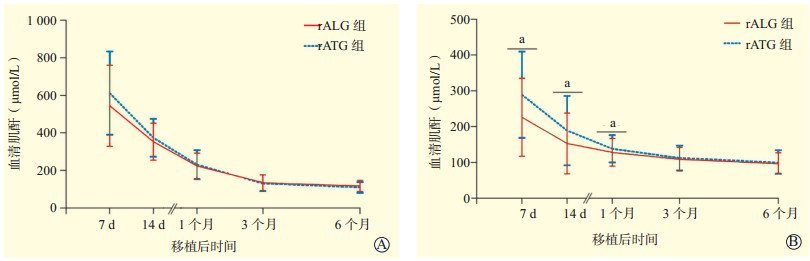
目的 比较不同T细胞多克隆抗体在公民逝世后器官捐献供肾肾移植中的临床疗效。 方法 回顾性分析行公民逝世后器官捐献供肾肾移植的691例供、受者的临床资料。根据用于诱导的T细胞多克隆抗体不同, 将受者分为兔抗人T细胞免疫球蛋白(rALG)组(414例)和兔抗人胸腺细胞免疫球蛋白(rATG)组(277例)。收集两组受者移植肾功能恢复情况, 包括受者肾移植术后移植肾功能延迟恢复(DGF)和急性排斥反应(AR)的发生情况、术后血清肌酐水平的变化; 收集肾移植术后受者和移植肾1年的生存率; 统计术后1年内不良反应的发生率; 按供者DGF风险评分分为5组, 统计各组受者的rALG和rATG的使用比例。 结果 rALG组和rATG组受者DGF发生率分别为14.5%(60/414)、11.9%(33/277), rALG组、rATG组受者DGF持续时间分别为(7±4)d、(12±7)d, 两组间差异无统计学意义(P > 0.05)。rALG组AR发生率为7.5%(31/414), 明显高于rATG组的4.0%(11/277)(P < 0.05)。两组受者肾移植术后6个月内血清肌酐均呈逐渐下降的趋势。供者DGF风险评分为0~15分的供肾肾移植中, rALG的使用比例显著高于rATG; 而供者DGF风险评分 > 16分的供肾肾移植中, rATG使用的比例显著高于rALG(P < 0.05)。rALG组和rATG组受者、移植肾1年存活率分别为99.8%和99.6%、98.1%和98.2%, 两组间差异均无统计学意义(均为P > 0.05)。rATG组受者急性肺水肿和白细胞减少的发生率显著高于rALG组(均为P < 0.05)。 结论 rALG和rATG均能有效降低公民逝世后器官捐献供肾肾移植术后DGF及AR发生率, 获得良好的临床效果, rATG诱导的肾移植受者白细胞减少和急性肺水肿发生率高于rALG。 Abstract:Objective To compare the clinical efficacy of different T lymphocyte polyclonal antibodies in renal transplantation from donor kidney of organ donation after citizen's death. Methods Clinical data of 691 donors and recipients undergoing renal transplantation from donor kidney of organ donation after citizen's death were retrospectively analyzed. According to different T lymphocyte polyclonal antibodies used for induction, all recipients were divided into the rabbit anti human T lymphocyte immunoglobulin (rALG) group (n=414) and rabbit anti human thymocyte immunoglobulin (rATG) group (n=277). The recovery of renal graft function in recipients of the two groups were collected, including the incidence of delayed graft function (DGF) and acute rejection (AR), and the changes of serum creatinine level after renal transplantation. The 1-year survival rate of the recipients and renal grafts was collected. The incidence of adverse effects within 1 year after operation was calculated. According to the DGF risk score of donors, all recipients were divided into 5 groups. The use proportion of rALG and rATG in the recipients of each group was calculated. Results The incidence of DGF in the recipients of rALG and rATG groups was 14.5% (60/414) and 11.9% (33/277), respectively. The duration of DGF in the recipients of rALG and rATG groups was (7±4) d and (12±7) d respectively, with no statistically significant difference between two groups (P > 0.05). The incidence of AR in the rALG group was 7.5% (31/414), significantly higher than 4.0% (11/277) in the rATG group (P < 0.05). The serum creatinine levels of recipients within 6 months after renal transplantation tended to gradually decline in both groups. In renal transplantation for donor kidney with a DGF risk score of 0-15, the use proportion of rALG was significantly higher than that of rATG. However, the use proportion of rATG was significantly higher than that of rALG in renal transplantation for donor kidney with a DGF risk score over 16 (P < 0.05). The 1-year survival rates of the recipients and renal grafts in the rALG and rATG groups were 99.8% and 99.6%, 98.1% and 98.2%, respectively. There was no significant difference between two groups (both P > 0.05). The incidence of acute pulmonary edema and leukopenia in the recipients of rATG group was significantly higher than that in the rALG group (both P < 0.05). Conclusions Both rALG and rATG can effectively reduce the incidence of DGF and AR and achieve good clinical efficacy after renal transplantation from donor kidney of organ donation after citizen's death. The incidence of leukopenia and acute pulmonary edema induced by rATG is higher than that by rALG in the renal transplant recipients. -
表 1 两组肾移植受者的并发症发生情况
Table 1. Occurrence of complications of renal transplant recipients in two groups [n(%)]
组别 n 感染 白细胞减少 血小板减少 急性肺水肿 rATG组 277 30
(10.8)37
(13.4)45
(16.2)7
(2.5)rALG组 414 39
(9.4)21
(5.1)42
(10.1)2
(0.5)P值 0.541 0.046 0.057 0.013 -
[1] Transplant Experts of the National Organ Donation and Transplantation Committee, Officers of National Health and Family Planning Commission, HUANG JF, et al. The new era of organ transplantation in China[J]. Chin Med J (Engl), 2016, 129(16):1891-1893. DOI: 10.4103/0366-6999.187865. [2] 庄杰, 代波, 巫丽娟, 等.乙型肝炎病毒携带者肾移植术后临床观察[J].器官移植, 2017, 8(1):66-72.DOI: 10.3969/j.issn.1674-7445.2017.01.013.ZHUANG J, DAI B, WU LJ, et al. Clinical observation of hepatitis B virus carriers after renal transplantation[J]. Organ Transplant, 2017, 8(1):66-72. DOI:10.3969/j.issn. 1674-7445.2017.01.013. [3] QUINTELLA AHDS, LASMAR MF, FABRETI-OLIVEIRA RA, et al. Delayed graft function, predictive factors, and 7-year outcome of deceased donor kidney transplant recipients with different immunologic profiles[J]. Transplant Proc, 2018, 50(3):737-742. DOI: 10.1016/j.transproceed.2018.02.007. [4] 阙宏亮, 韩志坚, 耿亮, 等.器官捐献肾移植受者近期预后的影响因素分析[J].南京医科大学学报(自然科学版), 2018, 38(5):616-621, 627.DOI: 10.7655/NYDXBNS20180509.QUE HL, HAN ZJ, GENG L, et al. Risk factors on the short-term outcome of DCD kidney transplanted recipients: a singlecenter study[J]. J Nanjing Med Univ, 2018, 38(5):616-621, 627. DOI: 10.7655/NYDXBNS20180509. [5] 周梅生, 朱有华, 王立明, 等.不同ATG在肾移植中的应用比较[J].临床泌尿外科杂志, 2008, 23(10):745-747. DOI: 10.3969/j.issn.1001-1420.2008.10.009.ZHOU MS, ZHU YH, WANG LM, et al. Comparison of different ATG in renal transplantation[J]. J Clin Urol, 2008, 23(10):745-747. DOI: 10.3969/j.issn.1001-1420.2008.10.009. [6] NAFAR M, DALILI N, POOR-REZA-GHOLI F, et al. The appropriate dose of thymoglobulin induction therapy in kidney transplantation[J]. Clin Transplant, 2017, 31(6). DOI: 10.1111/ctr.12977. [7] BOURDAGE JS, HAMLIN DM. Comparative polyclonal antithymocyte globulin and antilymphocyte/antilymphoblast globulin anti-CD antigen analysis by flow cytometry[J]. Transplantation, 1995, 59(8):1194-1200. doi: 10.1097/00007890-199504270-00020 [8] SONG T, YIN S, LI X, et al. Thymoglobulin vs. ATG-fresenius as induction therapy in kidney transplantation: a Bayesian network Meta-analysis of randomized controlled trials[J]. Front Immunol, 2020, 11:457. DOI: 10.3389/fimmu.2020.00457. [9] STYRC B, SOBOLEWSKI M, CHUDEK J, et al. Effectiveness and safety of two different antithymocyte globulins used in induction therapy in kidney transplant recipients: a single-center experience[J]. Clin Transplant, 2019, 33(10):e13680. DOI: 10.1111/ctr.13680. [10] 潘晓鸣, 薛武军, 田普训, 等.供者和供肾质量的评估及肾移植1 084例的临床经验总结[J].中华器官移植杂志, 2018, 11(39):645-650. DOI: 10.3969/j.issn.1674-7445.2020.03.010.PAN XM, XUE WJ, TIAN PX, et al. Quality Assessment of donor and donor kidney and clinical experience summary of 1 084 cases of renal transplantation[J]. Chin J Organ Transplant, 2018, 11(39):645-650. DOI: 10.3969/j.issn.1674-7445.2020.03.010. [11] HU XJ, ZHENG J, LI Y, et al. Prediction of kidney transplant outcome based on different DGF definitions in Chinese deceased donation[J]. BMC Nephrol, 2019, 20(1):409. DOI: 10.1186/s12882-019-1557-x. [12] MAUIYYEDI S, COLVIN RB. Humoral rejection in kidney transplantation: new concepts in diagnosis and treatment[J]. Curr Opin Nephrol Hypertens, 2002, 11(6):609-618. DOI: 10.1097/00041552-200211000-00007. [13] 陈莉萍, 莫春柏, 田军, 等.ATG-F单次大剂量与多次低剂量给药应用于肾移植诱导治疗的有效性和安全性[J].中华器官移植杂志, 2017, 38(11):665-670. DOI: 10.3760/cma.j.issn.0254-1785.2017.11.006.CHEN LP, MO CB, TIAN J, et al. Efficacy and safety of single high-dose versus multiple low-dose ATG-Fresenius induction in de novo renal transplantation[J]. Chin J Organ Transplant, 2017, 38(11):665-670. DOI: 10.3760/cma.j.issn.0254-1785.2017.11.006. [14] BRENNAN DC, DALLER JA, LAKE KD, et al. Rabbit antithymocyte globulin versus basiliximab in renal transplantation[J]. N Engl J Med, 2006, 355(19):1967-1977. DOI: 10.1056/NEJMoa060068. [15] 谢海瑞, 沈二霞, 李文益, 等.未甲基化CpG寡聚脱氧核苷酸对小鼠脾细胞产生免疫球蛋白E的抑制作用[J].中山大学学报(医学科学版), 2007, 28(4):361-366. http://www.cnki.com.cn/Article/CJFDTotal-ZSYK200704005.htmXIE HR, SHEN EX, LI WY, et al. Inhibitory effect of non-methylation CpG oligodeoxynucleotide on production of immunoglobulin E by murine splenocytes[J]. J Sun Yat-sen Univ(Med Sci), 2007, 28(4):361-366. http://www.cnki.com.cn/Article/CJFDTotal-ZSYK200704005.htm [16] NADERI N, ALAMDARI A, LESSAN-PEZESHKI M, et al. Risk factors for delayed graft function in deceased donor kidney transplantation; a potential preventive role for intraoperative thymoglobulin[J]. J Renal Inj Prev, 2019, 8(2): 157-163. DOI: 10.15171/jrip.2019.29. [17] 任吉忠, 闵志廉, 朱有华, 等.尸肾移植肾功能延迟恢复70例次总结[J].中华器官移植杂志, 1997, 18(2):103-104. DOI: 10.3760/cma.j.issn.0254-1785.1997.02.019.REN JZ, MIN ZL, ZHU YH, et al. Delayed graft function of cadaveric renal transplantation[J]. Chin J Organ Transplant, 1997, 18(2):103-104.DOI:10.3760/cma.j.issn. 0254-1785.1997.02.019. [18] MANNON RB. Delayed graft function: the AKI of kidney transplantation[J]. Nephron, 2018, 140(2):94-98. DOI: 10.1159/000491558. [19] YILMAZ M, SEZER TÖ, GÜNAY E, et al. Efficacy and safety of ATG-Fresenius as an induction agent in living-donor kidney transplantation[J]. Transplant Proc, 2017, 49(3):481-485. DOI: 10.1016/j.transproceed.2017.02.005. [20] POPOW I, LEITNER J, GRABMEIER-PFISTERSHAMMER K, et al. A comprehensive and quantitative analysis of the major specificities in rabbit antithymocyte globulin preparations[J]. Am J Transplant, 2013, 13(12):3103-3113. DOI: 10.1111/ajt.12514. [21] CREPIN T, CARRON C, ROUBIOU C, et al. ATG-induced accelerated immune senescence: clinical implications in renal transplant recipients[J]. Am J Transplant, 2015, 15(4):1028-1038. DOI: 10.1111/ajt.13092. [22] TERASAKO K, SATO K, SATO M, et al. The effect of different ATG preparations on immune recovery after allogeneic hematopoietic stem cell transplantation for severe aplastic anemia[J]. Hematology, 2010, 15(3):165-169. DOI: 10.1179/102453309X12583347113852. [23] GOGGINS WC, PASCUAL MA, POWELSON JA, et al. A prospective, randomized, clinical trial of intraoperative versus postoperative thymoglobulin in adult cadaveric renal transplant recipients[J]. Transplantation, 2003, 76(5):798-802. DOI: 10.1097/01.TP.0000081042.67285.91. -





 下载:
下载: