Establishment of a blood concentration detection system for everolimus in recipients after liver transplantation
-
摘要:
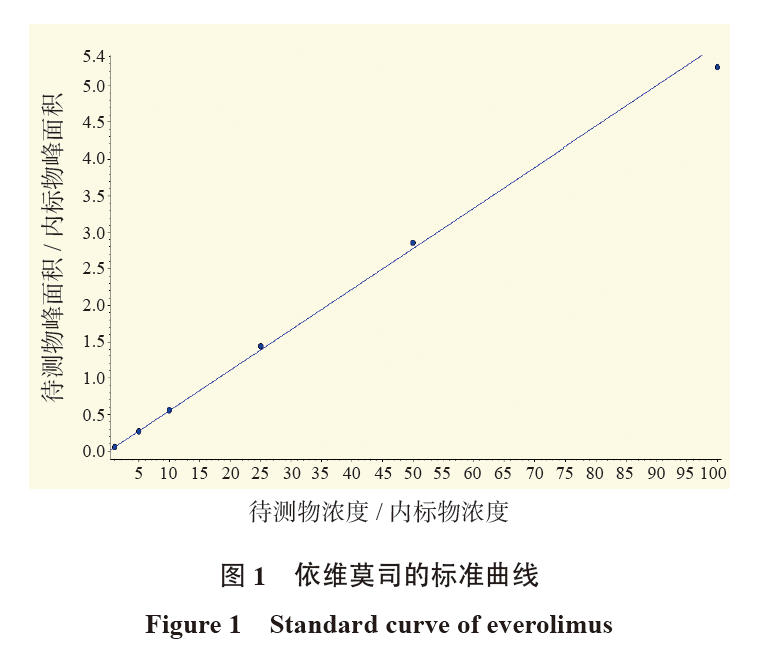
目的 建立测定肝移植受者全血中依维莫司浓度的超高效液相色谱-串联质谱法(UPLC-MS/MS)检测体系。 方法 用甲醇和硫酸锌沉淀样本蛋白质,以依维莫司-D4为内标物。采用Phenomenex Kinetex PFP色谱柱;流动相A:水(含有2 mmol/L甲酸铵和0.1%甲酸),流动相B:甲醇(含有2 mmol/L甲酸铵和0.1%甲酸),梯度洗脱,流速1 mL/min,柱温50 ℃,进样量1 μL。在正离子模式下,以多反应监测方式进行定量测定。该UPLC-MS/MS检测体系只需100 μL全血,无需复杂的样品制备即可达到足够的定量下限,总运行时间4.5 min内。采用依维莫司峰面积/依维莫司-D4峰面积(y)和依维莫司浓度/依维莫司-D4浓度(x)进行线性回归(1/ x2)分析,计算校准函数,分析其准确度和线性关系。并应用UPLC-MS/MS检测5例肝移植术后受者血样本的依维莫司血药谷浓度。 结果 依维莫司在血药浓度1~100 ng /mL范围内的质控准确度都在15%以内,线性关系良好(R2 > 0.990)。测定的5例肝移植受者血样本的依维莫司血药谷浓度为3.77~9.27 ng/mL。 结论 本研究建立的UPLC-MS/MS检测体系测定肝移植受者全血中依维莫司血药浓度准确度高、样品处理方法简便、检测时间短,适用于肝移植术后受者血样本中依维莫司的药物浓度监测。 -
关键词:
- 依维莫司 /
- 超高效液相色谱-串联质谱法(UPLC-MS/MS) /
- 高效液相色谱法(HPLC) /
- 肝移植 /
- 血药浓度 /
- 钙调磷酸酶抑制剂(CNI) /
- 哺乳动物雷帕霉素靶蛋白(mTOR) /
- 西罗莫司
Abstract:Objective To establish a detection system of ultra high performance liquid chromatography-tandem mass spectrometry (UPLC-MS/MS) for everolimus concentration in whole blood of liver transplant recipients. Methods The proteins of samples were precipitated with methanol and zinc sulfate, and everolimus-D4 was used as the internal standard. Phenomenex Kinetex PFP column was used. The mobile phase A was water (containing 2 mmol/Lammonium formate and 0.1% formic acid), and the mobile phase B was methanol (containing 2 mmol/L ammonium formate and 0.1% formic acid). The gradient elution was performed with the flow rate of 1 mL/min, the column temperature of 50 ℃ and the injection volume of 1 μL. The multi-reaction monitoring mode was used to quantitatively analyze with electrospray positive ionization. The UPLC-MS/MS detection system required only 100 μL of whole blood, and could achieve a sufficient lower limit of quantification without complicated sample preparation. The total running time was within 4.5 min. Linear regression (1/x2) analysis was performed using peak area of everolimus / peak area of everolimus-D4 (y) and concentration of everolimus/concentration of everolimus-D4 (x) to calculate the calibration function and analyze its accuracy and linear relationship. UPLC-MS/MS was used to detect the trough blood concentration of everolimus in blood samples of 5 recipients after liver transplantation. Results The accuracy of quality control was within 15%, and the linear relationship of everolimus was good in the blood concentration range of 1-100 ng /mL(R2 > 0.990). Trough blood concentration of everolimus measured in blood samples of 5 liver transplant recipients ranged from 3.77 to 9.27 ng/mL. Conclusions The detection system of UPLC-MS/MS in this study is suitable for monitoring the concentration of everolimus in whole blood of liver transplant recipients because of its high accuracy, simple sample processing method and short detection time. -
表 1 依维莫司标准曲线质控的准确度结果
Table 1. Accuracy results of quality control of everolimus standard curve
样本序号 待测物峰面积 理论浓度(ng/mL) 内标物峰面积 实测浓度(ng/mL) 准确度(%) STD1 5.13×103 1.00 1.01×105 1.00 100.0 STD2 2.33×104 5.00 8.63×104 4.85 97.0 STD3 5.05×104 10.00 9.21×104 9.75 97.5 STD4 1.39×105 25.00 8.88×104 27.60 110.4 STD5 2.62×105 50.00 9.09×104 50.80 101.6 STD6 5.00×105 100.00 9.46×104 93.20 93.2 L-QC 1.68×104 3.31 8.91×104 3.43 103.6 M-QC 7.81×104 13.00 1.07×105 13.00 100.0 H-QC 2.18×105 41.50 9.19×104 41.80 100.7 表 2 5例肝移植受者的依维莫司血药谷浓度检测结果
Table 2. Test results of everolimus trough blood concentration in 5 liver transplant recipients
样本序号 待测物峰面积 内标物峰面积 实测浓度(ng/mL) Sample-1 4.06×104 9.11×104 8.05 Sample-2 3.28×104 8.99×104 6.60 Sample-3 2.43×104 1.17×105 3.77 Sample-4 4.07×104 9.59×104 7.67 Sample-5 4.96×104 9.66×104 9.27 -
[1] KIM WR, LAKE JR, SMITH JM, et al. OPTN/SRTR 2013 annual data report: liver[J]. Am J Transplant, 2015, 15(Suppl 2): 1-28. DOI: 10.1111/ajt.13197. [2] 沈中阳, 谷川, 郑虹, 等. 临床肝脏移植20年回顾[J]. 中华危重病急救医学, 2019, 31(3): 269-280. DOI: 10.3760/cma.j.issn.2095-4352.2019.03.004.SHEN ZY, GU C, ZHENG H, et al. A twenty-year review of clinical liver transplantation[J]. Chin Crit Care Med, 2019, 31(3): 269-280. DOI: 10.3760/cma.j.issn.2095-4352.2019.03.004. [3] 赵东, 夏强. 肝移植相关领域的研究进展[J]. 国际消化病杂志, 2020, 40(2): 71-74. DOI: 10.3969/j.issn.1673-534X.2020.02.001.ZHAO D, XAI Q. Research progress in related fields of liver transplantation[J]. Int J Dig Dis, 2020, 40(2): 71-74. DOI: 10.3969/j.issn.1673-534X.2020.02.001. [4] LEMAITRE F, VETHE NT, D'AVOLIO A, et al. Measuring intracellular concentrations of calcineurin inhibitors: expert consensus from the International Association of Therapeutic Drug Monitoring and Clinical Toxicology Expert Panel[J]. Ther Drug Monit, 2020, 42(5): 665-670. DOI: 10.1097/FTD.0000000000000780. [5] HOŠKOVÁ L, MÁLEK I, KOPKAN L, et al. Pathophysiological mechanisms of calcineurin inhibitor-induced nephrotoxicity and arterial hypertension[J]. Physiol Res, 2017, 66(2): 167-180. DOI: 10.33549/physiolres.933332. [6] FAROUK SS, REIN JL. The many faces of calcineurin inhibitor toxicity-what the FK?[J]. Adv Chronic Kidney Dis, 2020, 27(1): 56-66. DOI: 10.1053/j.ackd.2019.08.006. [7] 中华医学会器官移植学分会. 器官移植免疫抑制剂临床应用技术规范(2019版)[J]. 器官移植, 2019, 10(3): 213-226. DOI: 10.3969/j.issn.1674-7445.2019.03.001.Branch of Organ Transplantation of Chinese Medical Association. Technical specifcation for clinical application of immunosuppressive agents in organ transplantation (2019 edition)[J]. Organ Transplant, 2019, 10(3): 213-226. DOI: 10.3969/j.issn.1674-7445.2019.03.001. [8] HASSKARL J. Everolimus[J]. Recent Results Cancer Res, 2018, 211: 101-123. DOI: 10.1007/978-3-319-91442-8_8. [9] ZOU Y, LI W, ZHOU J, et al. ERK inhibitor enhances everolimus efficacy through the attenuation of dNTP pools in renal cell carcinoma[J]. Mol Ther Nucleic Acids, 2019, 14: 550-561. DOI: 10.1016/j.omtn.2019.01.001. [10] CUCCHIARI D, RÍOS J, MOLINA-ANDUJAR A, et al. Combination of calcineurin and mTOR inhibitors in kidney transplantation: a propensity score analysis based on current clinical practice[J]. J Nephrol, 2020, 33(3): 601-610. DOI: 10.1007/s40620-019-00675-2. [11] GEDALY R, DE STEFANO F, TURCIOS L, et al. mTOR inhibitor everolimus in regulatory T cell expansion for clinical application in transplantation[J]. Transplantation, 2019, 103(4): 705-715. DOI: 10.1097/TP.0000000000002495. [12] BENDTSEN MAF, GRIMM D, BAUER J, et al. Hypertension caused by lenvatinib and everolimus in the treatment of metastatic renal cell carcinoma[J]. Int J Mol Sci, 2017, 18(8): 1736. DOI: 10.3390/ijms18081736. [13] TAN PS, MUTHIAH MD, KOH T, et al. Asian Liver Transplant Network clinical guidelines on immunosuppressionin liver transplantation[J]. Transplantation, 2019, 103(3): 470-480. DOI: 10.1097/TP.0000000000002532. [14] CILLO U, DE CARLIS L, DEL GAUDIO M, et al. Immunosuppressive regimens for adult liver transplant recipients in real-life practice: consensus recommendations from an Italian Working Group[J]. Hepatol Int, 2020, 14(6): 930-943. DOI: 10.1007/s12072-020-10091-5. [15] RUBÍN SUÁREZ A, BILBAO AGUIRRE I, FERNÁNDEZ-CASTROAGUDIN J, et al. Recommendations of everolimususe in liver transplant[J]. Gastroenterol Hepatol, 2017, 40(9): 629-640. DOI: 10.1016/j.gastrohep.2017.05.008. [16] LIN M, MITTAL S, SAHEBJAM F, et al. Everolimus with early withdrawal or reduced-dose calcineurin inhibitors improves renal function in liver transplant recipients: a systematic review and Meta-analysis[J]. Clin Transplant, 2017, 31(2): e12872. DOI: 10.1111/ctr.12872. [17] YU L, CHEN X, WANG L, et al. The sweet trap in tumors: aerobic glycolysis and potential targets for therapy[J]. Oncotarget, 2016, 7(25): 38908-38926. DOI: 10.18632/oncotarget.7676. [18] FERRÍN G, GUERRERO M, AMADO V, et al. Activation of mTOR signaling pathway in hepatocellular carcinoma[J]. Int J Mol Sci, 2020, 21(4): 1266. DOI: 10.3390/ijms21041266. [19] VAN GELDER T, FISCHER L, SHIHAB F, et al. Optimizing everolimus exposure when combined with calcineurin inhibitors in solid organ transplantation[J]. Transplant Rev (Orlando), 2017, 31(3): 151-157. DOI: 10.1016/j.trre.2017.02.007. [20] MABASA VH, ENSOM MH. The role of therapeutic monitoring of everolimus in solid organ transplantation[J]. Ther Drug Monit, 2005, 27(5): 666-676. DOI: 10.1097/01.ftd.0000175911.70172.2e. [21] FALKOWSKI S, WOILLARD JB. Therapeutic drug monitoring of everolimus in oncology: evidences and perspectives[J]. Ther Drug Monit, 2019, 41(5): 568-574. DOI: 10.1097/FTD.0000000000000628. [22] STROBBE G, PANNIER D, SAKJI I, et al. Advantages of everolimus therapeutic drug monitoring in oncology when drug-drug interaction is suspected: a case report[J]. J Oncol Pharm Pract, 2020, 26(7): 1743-1749. DOI: 10.1177/1078155220904761. [23] GOIRAND F, ROYER B, HULIN A, et al. Level of evidence for therapeutic drug monitoring of everolimus[J]. Therapie, 2011, 66(1): 57-61. DOI: 10.2515/therapie/2010025. [24] VENTURA-AGUIAR P, CAMPISTOL JM, DIEKMANN F. Safety of mTOR inhibitors in adult solid organ transplantation[J]. Expert Opin Drug Saf, 2016, 15(3): 303-319. DOI: 10.1517/14740338.2016.1132698. [25] ARENA C, BIZZOCA ME, CAPONIO VCA, et al. Everolimus therapy and side-effects: a systematic review and Meta-analysis[J]. Int J Oncol, 2021, 59(1): 54. DOI: 10.3892/ijo.2021.5234. [26] YEE ML, TAN HH. Use of everolimus in liver transplantation[J]. World J Hepatol, 2017, 9(23): 990-1000. DOI: 10.4254/wjh.v9.i23.990. [27] NOGUERAS LÓPEZ F, ABELLAN ALFOCEA P, ORTEGA SUAZO EJ, et al. Impact of everolimus-based immunosuppression on renal function in liver transplant recipients[J]. Transplant Proc, 2020, 52(2): 556-558. DOI: 10.1016/j.transproceed.2019.12.012. [28] KOVARIK JM, TEDESCO H, PASCUAL J, et al. Everolimus therapeutic concentration range defined from a prospective trial with reduced-exposure cyclosporine in de novo kidney transplantation[J]. Ther Drug Monit, 2004, 26(5): 499-505. DOI: 10.1097/00007691-200410000-00007. [29] SHIPKOVA M, HESSELINK DA, HOLT DW, et al. Therapeutic drug monitoring of everolimus: a consensus report[J]. Ther Drug Monit, 2016, 38(2): 143-169. DOI: 10.1097/FTD.0000000000000260. [30] KLAWITTER J, NASHAN B, CHRISTIANS U. Everolimus and sirolimus in transplantation-related but different[J]. Expert Opin Drug Saf, 2015, 14(7): 1055-1070. DOI: 10.1517/14740338.2015.1040388. [31] JASIAK NM, PARK JM. Immunosuppression in solid-organ transplantation: essentials and practical tips[J]. Crit Care Nurs Q, 2016, 39(3): 227-240. DOI: 10.1097/CNQ.0000000000000117. [32] SATOH S, MIURA M. Therapeutic drug monitoring of immunosuppressive drugs[J]. Rinsho Byori, 2016, 64(12): 1381-1389. [33] MILLÁN O, WIELAND E, MARQUET P, et al. Pharmacodynamic monitoring of mTOR inhibitors[J]. Ther Drug Monit, 2019, 41(2): 160-167. DOI: 10.1097/FTD.0000000000000616. [34] ZHANG M, TAJIMA S, SHIGEMATSU T, et al. Development and validation of an LC-MS/MS Method to simultaneously measure tacrolimus and everolimus concentrations in kidney allograft biopsies after kidney transplantation[J]. Ther Drug Monit, 2021, DOI: 10.1097/FTD.0000000000000912[Epubaheadofprint]. [35] ANTUNES NJ, KIPPER K, COUCHMAN L, et al. Simultaneous quantification of cyclosporin, tacrolimus, sirolimus and everolimus in whole blood by UHPLC-MS/MS for therapeutic drug monitoring[J]. Biomed Chromatogr, 2021, 35(6): e5071. DOI: 10.1002/bmc.5071. [36] 洪顺福, 吴国兰, 郑运亮, 等. 在线固相萃取LC-MS/MS法测定健康人体全血中依维莫司浓度及在药代动力学研究中的应用[J]. 药物分析杂志, 2016, 36(10): 1778-1784. DOI: 10.16155/j.0254-1793.2016.10.11.HONG FS, WU GL, ZHENG YL, et al. Determination of everolimus in whole blood by online SPE LC-MS/MS system and its application in pharmacokinetic study[J]. Chin J Pharm Anal, 2016, 36(10): 1778-1784. DOI: 10.16155/j.0254-1793.2016.10.11. [37] 翟晓慧, 刘晓雪, 陆佳倩, 等. LC-MS/MS法同时分析血液中几种免疫抑制剂浓度[J]. 中国医院药学杂志, 2019, 39(8): 774-780. DOI: 10.13286/j.cnki.chinhosppharmacyj.2019.08.02.ZHAI XH, LIU XX, LU JQ, et al. Establishment of LC-MS/MS assay for the determination of blood immunosuppressive agents level simultaneously[J]. Chin J Hosp Pharm, 2019, 39(8): 774-780. DOI: 10.13286/j.cnki.chinhosppharmacyj.2019.08.02. -





 下载:
下载: