Clinical efficacy of ABO-incompatible living donor liver transplantation: a Meta analysis
-
摘要:
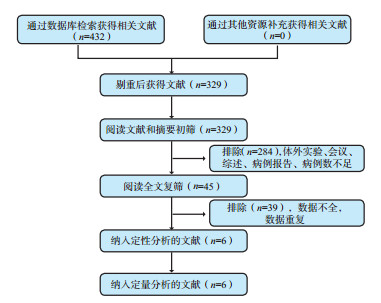
目的 系统评价ABO血型不合活体肝移植(ABO-I LDLT)术后的疗效,并与ABO血型相合活体肝移植(ABO-C LDLT)相比较。 方法 系统检索国内外多个数据库,收集关于ABO-I LDLT及ABO-C LDLT疗效对比的文献。根据标准筛选文献,并进行文献质量评价,提取数据。采用Rev Man 5.3软件,应用随机效应模型或固定效应模型进行Meta分析。 结果 检索文献432篇,按纳入标准筛选共纳入6篇英文文献。Meta分析结果表明ABO-I LDLT组与ABO-C LDLT组的受体及移植物术后1、3、5年存活率和排斥反应发生率差异均无统计学意义(均为P≥0.05);ABO-I LDLT组术后胆道并发症发生率和肝动脉栓塞发生率均高于ABO-C LDLT组,差异均有统计学意义[比值比(OR)=2.08,95%可信区间(CI)1.25~3.45,P=0.005;OR=2.24,95%CI 1.03~4.89,P=0.04)。 结论 与ABO-C LDLT相比,ABO-I LDLT疗效稍差,但仍是一种可供选择的治疗终末期肝病的有效手段。 Abstract:Objective To systematically evaluate the clinical efficacy of ABO-incompatible living donor liver transplantation (ABO-I LDLT) and compare with ABO-compatible LDLT (ABO-C LDLT). Methods A systematic search of multiple databases at home and abroad was conducted to retrieve the literatures related to the statistical comparison of clinical efficacy between ABO-I LDLT and ABO-C LDLT. The literature screening was conducted, the quality of literatures was evaluated and data extraction was performed. Using Rev Man 5.3 software, a Meta-analysis was performed by random effect model or fixed effect model. Results A total of 432 articles were searched, and 6 articles published in English were eventually included according to the inclusion criteria. The Meta-analysis demonstrated that there was no significant difference in the postoperative 1-, 3- and 5-year survival rate of the recipients and grafts and the incidence of rejection responses between the ABO-I LDLT and ABO-C LDLT groups (all P≥0.05). The incidence of postoperative biliary complications and hepatic artery embolization in the ABO-I LDLT group was significantly higher than that in the ABO-C LDLT group [odds ratio (OR) =2.08, 95% confidence interval (CI) 1.25-3.45, P=0.005; OR=2.24, 95%CI 1.03-4.89, P=0.04]. Conclusions Compared with the ABO-C LDLT, ABO-I LDLT yields lower clinical efficacy, whereas it is still an effective method for the treatment of end-stage liver disease. -
图 8 ABO-I LDLT组与ABO-C LDLT组胆道并发症发生率比较
Figure 8. Figure 8 Comparison of the incidence of biliary complications between ABO-I LDLT group and ABO-C LDLT group
表 1 纳入文献的基本信息及质量评分
Table 1. Basic information and scores of quality assessment of the included articles
研究作者 年份 国家或地区 文献类型 病例收集年份 ABO-I LDLT组 ABO-C LDLT组 NOS评分(分) 总例数 受体年龄 总例数 受体年龄 Song GW, et al[3] 2016 韩国 回顾性队列研究 2008~2013 235 25~68岁 1 301 19~70岁 8 Lee CF, et al[4] 2015 中国台湾 回顾性队列研究 2006~2013 046 19~67岁 0340 18~70岁 8 Kim JM, et al[5] 2016 韩国 回顾性队列研究 2010~2013 47 22~65岁 94 20~68岁 8 Bang JB, et al[6] 2016 韩国 回顾性队列研究 2008~2014 27 31~65岁 114 - 7 Egawa H, et al[10] 2004 日本 回顾性队列研究 1990~2000 66 3个月~55岁 461 1个月~68岁 7 Kim JD, et al[11] 2016 韩国 回顾性队列研究 2011~2014 25 35~66岁 157 44岁~57岁 8 -代表研究中未具体提及或难以提取 -
[1] CHEN CL, KABILING CS, CONCEJERO AM. Why does living donor liver transplantation flourish in Asia?[J]. Nat Rev Gastroenterol Hepatol, 2013, 10(12): 746-751. DOI: 10.1038/nrgastro.2013.194. [2] 蒋文涛, 沈中阳. 成人活体肝移植的现状和进展[J/CD]. 实用器官移植电子杂志, 2017, 5(4): 258-263. DOI: 10.3969/j.issn.2095-5332.2017.04.003.JIANG WT, SHEN ZY. Current status and progress of adult living donor liver transplantation [J/CD]. Pract J Organ Transplant (Electr Vers), 2017, 5(4): 258-263. DOI: 10.3969/j.issn.2095-5332.2017.04.003. [3] SONG GW, LEE SG, HWANG S, et al. ABO-incompatible adult living donor liver transplantation under the desensitization protocol with rituximab[J]. Am J Transplant, 2016, 16(1): 157-170. DOI: 10.1111/ajt.13444. [4] LEE CF, CHENG CH, WANG YC, et al. Adult living donor liver transplantation across ABO-incompatibility[J]. Medicine (Baltimore), 2015, 94(42): e1796. DOI: 10.1097/MD.0000000000001796. [5] KIM JM, KWON CH, JOH JW, et al. Case-matched comparison of ABO-incompatible and ABO-compatible living donor liver transplantation[J]. Br J Surg, 2016, 103(3): 276-283. DOI: 10.1002/bjs.10048. [6] BANG JB, KIM BW, KIM YB, et al. Risk factor for ischemic-type biliary lesion after ABO-incompatible living donor liver transplantation[J]. World J Gastroenterol, 2016, 22(30): 6925-6935. DOI: 10.3748/wjg.v22.i30.6925. [7] STEWART ZA, LOCKE JE, MONTGOMERY RA, et al. ABO-incompatible deceased donor liver transplantation in the United States: a national registry analysis[J]. Liver Transpl, 2009, 15(8): 883-893. DOI: 10.1002/lt.21723. [8] RUMMLER S, BAUSCHKE A, BAERTHEL E, et al. ABO-incompatible living donor liver transplantation in focus of antibody rebound[J]. Transfus Med Hemother, 2017, 44(1):46-51. DOI: 10.1159/000450792. [9] 曾宪涛, 刘慧, 陈曦, 等.Meta分析系列之四:观察性研究的质量评价工具[J].中国循证心血管医学杂志, 2012, 4(4): 297-299. DOI: 10.3969/j.1674-4055.2012.04.004.ZENG XT, LIU H, CHEN X, et al. Meta analysis series four: quality assessment tools for observational research[J]. Chin J Evid Base Cardiovascul Med, 2012, 4(4): 297-299. DOI: 10.3969/j.1674-4055.2012.04.004. [10] EGAWA H, OIKE F, BUHLER L, et al. Impact of recipient age on outcome of ABO-incompatible living-donor liver transplantation[J]. Transplantation, 2004, 77(3): 403-411. doi: 10.1097/01.TP.0000110295.88926.5C [11] KIM JD, CHOI DL, KIM SG, et al. Single-center experience of ABO-incompatible living-donor liver transplantation with a new simplified intravenous immunoglobulin protocol: a propensity score-matching analysis[J]. Transplant Proc, 2016, 48(4): 1134-1138. DOI: 10.1016/j.transproceed.2016.02.040. [12] RUMMLER S, BAUSCHKE A, BÄRTHEL E, et al. Current techniques for ABO-incompatible living donor liver transplantation[J]. World J Transplant, 2016, 6(3): 548-555. DOI: 10.5500/wjt.v6.i3.548. [13] HAGA H, EGAWA H, FUJIMOTO Y, et al. Acute humoral rejection and C4d immunostaining in ABO blood type-incompatible liver transplantation[J]. Liver Transpl, 2006, 12(3): 457-464. doi: 10.1002/(ISSN)1527-6473 [14] EGAWA H, UMESHITA K, UEMOTO S. Optimal dosage regimen for rituximab in ABO-incompatible living donor liver transplantation[J]. J Hepatobiliary Pancreat Sci, 2017, 24(2): 89-94. DOI: 10.1002/jhbp.419. [15] RAVN V, DABELSTEEN E. Tissue distribution of histo-blood group antigens[J]. APMIS, 2000, 108(1):1-28. doi: 10.1034/j.1600-0463.2000.d01-1.x [16] NISHIDA S, NAKAMURA N, KADONO J, et al. Intrahepatic biliary strictures after liver transplantation[J]. J Hepatobiliary Pancreat Surg, 2006, 13(6): 511-516. doi: 10.1007/s00534-005-1081-1 [17] LEE SD, KIM SH, KONG SY, et al. ABO-incompatible living donor liver transplantation without graft local infusion and splenectomy[J]. HPB(Oxford), 2014, 16(9): 807-813. DOI: 10.1111/hpb.12215. [18] LEE DW, JO HH, ABDULLAH J, et al. Endoscopic management of anastomotic strictures after liver transplantation[J]. Clin Endosc, 2016, 49(5): 457-461. doi: 10.5946/ce.2016.130 [19] CHENG YF, OU HY, YU CY, et al. Interventional radiology in living donor liver transplant[J]. World J Gastroenterol, 2014, 20(20): 6221-6225. DOI: 10.3748/wjg.v20.i20.6221. [20] NACIF LS, BERNARDO WM, BERNARDO L, et al. Endoscopic treatment of post-liver transplantation anastomotic biliary stricture: systematic review and Meta-analysis[J]. Arq Gastroenterol, 2014, 51(3): 240-249. doi: 10.1590/S0004-28032014000300014 [21] WU J, YE S, XU X, et al. Recipient outcomes after ABO-incompatible liver transplantation: a systematic review and Meta-analysis[J]. PLoS One, 2011, 6(1): e16521. DOI: 10.1371/journal.pone.0016521. [22] 董红锰, 代扬, 张欣雪, 等.ABO血型不合肝移植疗效的Meta分析[J].器官移植, 2016, 7(5): 370-377. DOI: 10.3969/j.issn.1674-7445.2016.05.008.DONG HM, DAI Y, ZHANG XX, et al. Meta-analysis of therapeutic effects on patients with ABO-incompatibility liver transplantation[J]. Organ Transplant, 2016, 7(5): 370-377. DOI: 10.3969/j.issn.1674-7445.2016.05.008. -





 下载:
下载: